Zaragozic acid A
- CAS NO.:142561-96-4
- Empirical Formula: C35H46O14
- Molecular Weight: 690.73
- MDL number: MFCD00868072
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 14:58:10
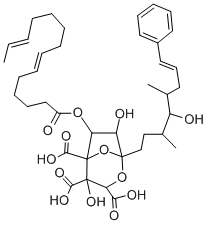
What is Zaragozic acid A?
Chemical properties
White to light tan solid. Soluble in ethanol (10 mg/ml), methanol, DMF, DMSO (10 mg/ml), and water (moderate).
The Uses of Zaragozic acid A
Zaragozic acid A is the major metabolite of a class of unusual bicyclic tricarboxylic acids, produced by a number of fungi in the genera Curvularia, Exserohilum, Setosphaeria and others, discovered at Merck and Glaxo in the early 1990s. In nature, zaragozic acid A acts as broad spectrum antifungal but its mode of action as an inhibitor of squalene synthase, involved in sterol biosynthesis, lead to investigation as a cholesterol lowering agent. Zaragozic acid A prepared by BioAustralis is presented as the free acid rather than the tri-sodium salt to avoid stability problems associated with hydrolysis of the salt.
What are the applications of Application
Zaragozic Acid A is a squalene synthetase inhibitor and fungal metabolite containing a novel 4,6,7-trihydroxy-2,8-dioxobicyclo[3.2.1]octane-3,4,5-tricarboxylic acid core.
Definition
ChEBI: Zaragozic acid A is a polyketide isolated from fungi that is a potent inhibitor of fungal and mammalian squalene synthase. It has a role as an EC 2.5.1.21 (squalene synthase) inhibitor and a fungal metabolite. It is a tricarboxylic acid, an acetate ester, a cyclic ketal, an oxabicycloalkane, a tertiary alcohol and a polyketide.
What are the applications of Application
Zaragozic Acid A is a squalene synthetase inhibitor. it targeted squalene synthase and lowered the cholesterol levels in vivo of marmosets.
Biological Activity
Zaragozic acid A is an inhibitor of acute liver cholsterol synthesis (50% inhibition dose 200 μg/ kg) with Ki value of 78pM. It inhibited cholesterol synthesis in Hep G2 cells.
in vitro
zaragozic acid a, as well as it two analogs zaragozic acid b and c were discovered from an unidentified sterile fungal culture, spororniella intermedia, and leptodontium elatius, respectively. zaragozic acid a, b and c were reported to be three potent competitive inhibitors of rat liver squalene synthase with ki values of 78 pm, 29 pm, and 45 pm, respectively. in addition, zaragozic acid a, b and c could also inhibite cholesterol synthesis in hep g2 cells [1].
in vivo
in the mouse model, zaragozic acid a was found to be an inhibitor of acute hepatic cholesterol synthesis (50% inhibitory dose of 200 μg/kg). the in-vivo inhibition of squalene synthase was accompanied by an accumulation of label from [3hjmevalonate into farnesyl diphosphate, farnesol, as well as organic acids. in summary, these data showed the zaragozic acids were a new class of therapeutic agents with potential for the hypercholesterolemia treatment [1].
References
[1] bergstrom jd,kurtz mm,rew dj,amend am,karkas jd,bostedor rg,bansal vs,dufresne c,vanmiddlesworth fl,hensens od, et al. zaragozic acids: a family of fungal metabolites that are picomolar competitive inhibitors of squalene synthase. proc natl acad sci u s a.1993 jan 1;90(1):80-4.
Properties of Zaragozic acid A
| Boiling point: | 850.1±65.0 °C(Predicted) |
| alpha | D22 +18.3° (c = 0.60 in CHCl3); D25 +37° (c = 1.29 in methanol) |
| Density | 1.35±0.1 g/cm3(Predicted) |
| storage temp. | −20°C |
| solubility | Soluble in DMSO |
| form | solid |
| pka | 1.47±0.70(Predicted) |
| color | white to light tan |
| CAS DataBase Reference | 142561-96-4 |
Safety information for Zaragozic acid A
Computed Descriptors for Zaragozic acid A
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 2-Bromo-5-Iodopyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 2-Hydroxy-5-methoxybenzoic acid 2-Cyanopyrimidine Xanthene-9-carboxylic acid 4,4-Diethoxybutanenitrile Methyl 4-amino-2,3-difluoro-5-nitrobenzoate 5-fluoro-1,3-benzodioxole 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 4-Chloro-2-methyl quinoline 2-Amino-4-phenyl-thiazole 2-(Chloromethyl) quinazolin-4(3H)-oneRelated products of tetrahydrofuran


You may like
-
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
