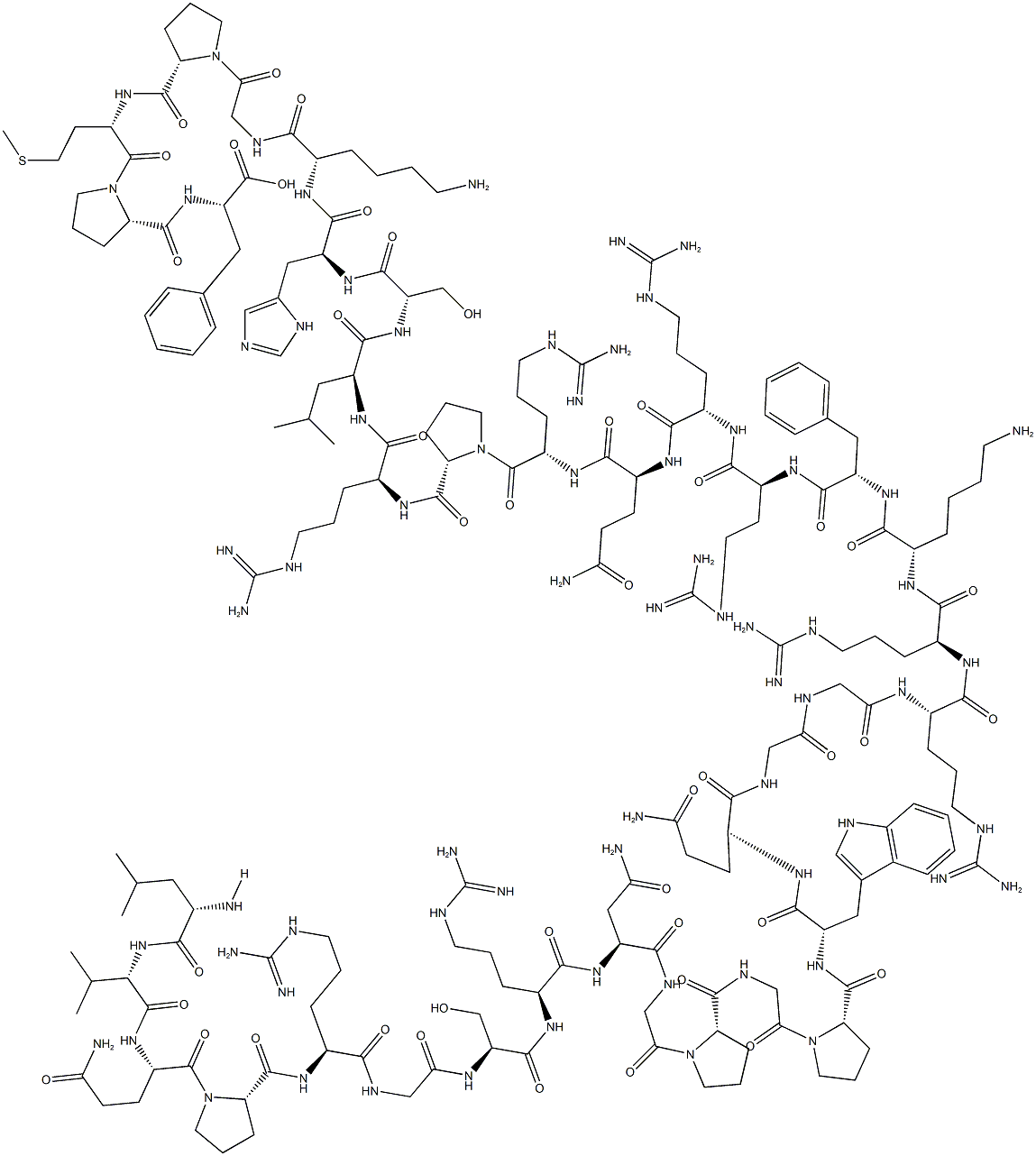
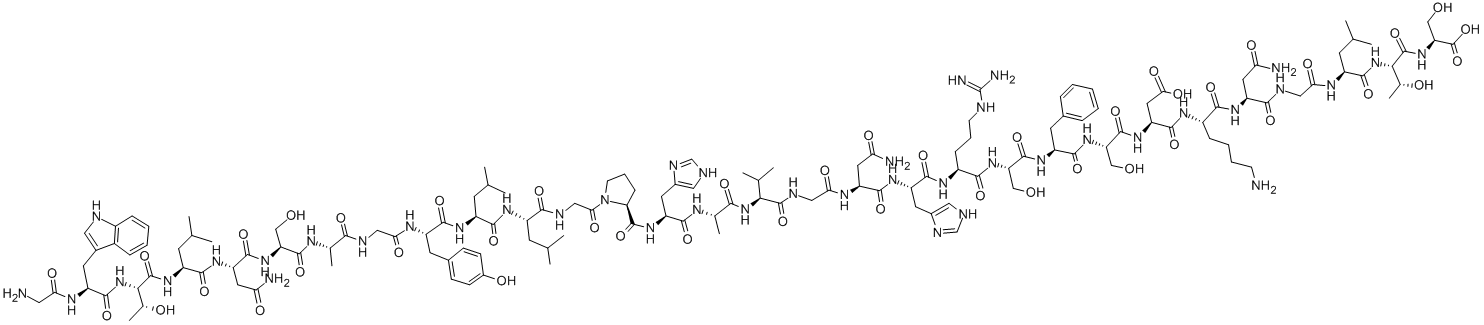
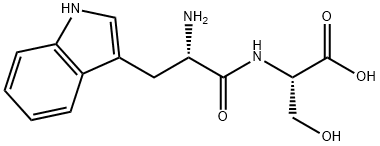
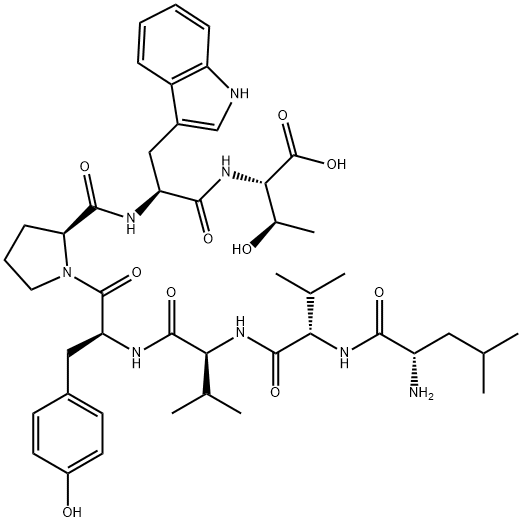
spinorphin
- CAS NO.:137201-62-8
- Empirical Formula: C45H64N8O10
- Molecular Weight: 877.04
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-22 18:57:36
What is spinorphin?
The Uses of spinorphin
Spinorphin is a novel neuropeptide, which exhibits complete inhibition of agonist-induced nociception.It is peptide analogs with branched amino acid probes and their therapeutic use.
What are the applications of Application
Spinorphin is an endogenous factor that exhibits inhibitory effects on enkephalin-degrading enzymes
Definition
ChEBI: Spinorphin is an oligopeptide.
Biological Activity
spinorphin is an endogenous inhibitor of enkephalin-degrading enzymes (aminopeptidase, dipeptidyl aminopeptidase iii, angiotensin-converting enzyme and enkephalinase) [1]. also, it is a potent antagonist of human p2x3 receptor and the n-formylpeptide receptor subtype fpr with ic50 value of 8.3 pm for p2x3 receptor[2][3].enkephalin degrading enzymes are a series of enzymes that hydrolyze enkephalins, which play an important role in management of blood pressure, pain, hypertension and cardiovascular diseases.in human embryonic kidney (hek) 293 cells transfected with mouse fpr, spinorphin induced a typical rise in the intracellular ca2+ concentration with ec50 of 128 μm. also, in normal mouse neutrophils, spinorphin induced calcium flux in a dose-dependent way. while the neutrophils from mice deficient in the fmlf receptor subtype fpr didn’t response [3].in mice, intrathecal administration of spinorphin inhibited the allodynia induced by intrathecal nociceptin in a dose-dependent way. furthermore, spinorphin enhanced the inhibitory effect of enkephalin on allodynia induced by nociceptin [4].
References
[1]. nishimura k, hazato t. isolation and identification of an endogenous inhibitor of enkephalin-degrading enzymes from bovine spinal cord. biochem biophys res commun, 1993, 194(2): 713-719.
[2]. jung ky, moon hd, lee ge, et al. structure-activity relationship studies of spinorphin as a potent and selective human p2x(3) receptor antagonist. j med chem, 2007, 50(18): 4543-4547.
[3]. liang ts, gao jl, fatemi o, et al. the endogenous opioid spinorphin blocks fmet-leu-phe-induced neutrophil chemotaxis by acting as a specific antagonist at the n-formylpeptide receptor subtype fpr. j immunol, 2001, 167(11): 6609-6614.
[4]. honda m, okutsu h, matsuura t, et al. spinorphin, an endogenous inhibitor of enkephalin-degrading enzymes, potentiates leu-enkephalin-induced anti-allodynic and antinociceptive effects in mice. jpn j pharmacol, 2001, 87(4): 261-267.
Properties of spinorphin
| storage temp. | Desiccate at -20°C |
| solubility | Soluble to 1 mg/ml in H2O |
| form | solid |
| color | White |
| Water Solubility | Soluble to 1 mg/ml in water |
| Stability: | Hygroscopic |
Safety information for spinorphin
Computed Descriptors for spinorphin
New Products
5-Chlorothiophene-2-carboxylic acid 2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-3-methyl xanthine Maleic hydrazide 6-Methyl-2-(p-tolyl)imidazo[1,2-a]pyridine 3,4-Dihydroxyacetophenone 3,4-Dimethoxyacetophenone 4-Nitroacetophenone DL-Glutamic acid 3-Nitroacetophenone 4'-Chloro-3'-nitroacetophenone 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 1-Boc-4-cyanopiperidine 2-Fluoro-6-iodobenzoic acid 3-Pyridineacrylic acid 3-Hydroxy-2-methylbenzoic acid 1,3-Dibromo-5-fluorobenzene 2-((2,6-Difluorobenzyl)(ethoxycarbonyl)amino)-4-((dimethylamino)methyl)-5-(4-nitrophenyl)thiophene-3-carboxylic acid [N(E)]-N-Ethylidene-2-methyl-2-propanesulfinamide 4-Isopropyl Thiophenol 6-(chloromethyl)pteridine-2,4-diamine monohydrochloride 2-Chlorophenyl diphenyl chloromethaneRelated products of tetrahydrofuran
You may like
-
 586-37-8 98+View Details
586-37-8 98+View Details
586-37-8 -
 1-Boc-Azetidine-3-yl-methanol 98+View Details
1-Boc-Azetidine-3-yl-methanol 98+View Details
142253-56-3 -
 4-Nitroacetophenone 100-19-6 98+View Details
4-Nitroacetophenone 100-19-6 98+View Details
100-19-6 -
 617-65-2 DL-Glutamic acid 98+View Details
617-65-2 DL-Glutamic acid 98+View Details
617-65-2 -
 1197-09-7' 98+View Details
1197-09-7' 98+View Details
1197-09-7' -
 3,4-Dimethoxyacetophenone 98+View Details
3,4-Dimethoxyacetophenone 98+View Details
1131-62-0 -
 4-Acetylbiphenyl 92-91-1 98+View Details
4-Acetylbiphenyl 92-91-1 98+View Details
92-91-1 -
 99-93-4 98+View Details
99-93-4 98+View Details
99-93-4