Meptazinol
- CAS NO.:54340-58-8
- Empirical Formula: C15H23NO
- Molecular Weight: 233.35
- MDL number: MFCD00864339
- EINECS: 259-109-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-02-27 17:19:27
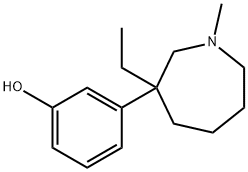
What is Meptazinol?
Originator
Meptid,Wyeth,UK,1983
The Uses of Meptazinol
Analgesic.
Definition
ChEBI: 3-(3-ethyl-1-methyl-3-azepanyl)phenol is a member of azepanes.
Manufacturing Process
2-(m-Methoxyphenyl)butyronitrile in dry ether was added to a stirred suspension of sodium amide in liquid ammonia. The mixture was stirred for 30 minutes then ethyl-4-iodobutyrate (99.25 g, 0.4 mol) in dry ether (200 ml) was added dropwise. The mixture was stirred at the temperature of refluxing liquid ammonia for 5 hours. Ammonium chloride (10 g) was added and the mixture allowed to warm to room temperature. Water (300 ml) was added, the organic layer separated, washed with water, 2 N sulfuric acid and water. After drying over magnesium sulfate and removing the ether, the product was distilled yielding ethyl 5-cyano-5-(mmethoxyphenyl)heptanoate.
That material was hydrogenated in cyclohexane using a Raney nickel catalyst. The product after distillation was recrystallized from ethyl acetate affording 10.0 g of 6-ethyl-(m-methoxyphenyl)hexahydro-2H-azepin-2one, MP 87°C to 88°C.
The azepinone (9.1 g) in dry tetrahydrofuran (50 ml) and ether (50 ml) was added dropwise to a stirred suspension of aluminum lithium hydride (7.5 g) in dry ether (50 ml). After heating under reflux for 3 hours the reaction mixture was worked up and distilled yielding 7.66 g of a compound which was a colorless oil, BP 108°C to 110°C/0.01 mm.
That product was then heated under reflux with 50% hydrobromic acid for 1.5
hours. The reaction mixture was evaporated to dryness and reevaporated with
three portions of propan-2-ol. The oil obtained was dissolved in propan-2-ol
and diluted with ether. 3-Ethyl-3-(m-hydroxyphenyl)hexahydro-1H-azepine
was obtained. That material in turn was reductively methylated by
hydrogenation in the presence of formaldehyde in absolute ethanol solution to
give 3-ethyl-3-(m-methoxyphenyl)-1-methylhexahydro-1H-azepine.
The methoxy group was converted to a hydroxy group by refluxing with 80%
HBr giving meptazinol hydrobromide.
Therapeutic Function
Analgesic
Clinical Use
Opioid analgesic used for moderate to severe pain
Drug interactions
Potentially hazardous interactions with other drugs
Antidepressants: possible CNS excitation or
depression with MAOIs - avoid; possible CNS
excitation or depression with moclobemide; possibly
increased sedative effects with tricyclics.
Antihistamines: increased sedative effects with
sedating antihistamines.
Antipsychotics: enhanced hypotensive and sedative
effects.
Dopaminergics: avoid with selegiline.
Nalmefene: avoid concomitant use.
Sodium oxybate: enhanced effect of sodium oxybate
- avoid
Metabolism
Meptazinol is extensively metabolised in the liver and is excreted mainly in the urine as the glucuronide conjugate. Less than 10% of a dose has been recovered from the faeces.
Properties of Meptazinol
| Melting point: | 127-133°C |
| Boiling point: | 354.8±35.0 °C(Predicted) |
| Density | 0.997 |
| pka | 9.95±0.10(Predicted) |
Safety information for Meptazinol
Computed Descriptors for Meptazinol
New Products
Pentadecanoic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 2-Bromo-5-Iodopyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile Tetrahydropyranyl-4-acetic acid Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside 5-fluoro-1,3-benzodioxole 2-Bromo-6-fluoroaniline 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-oneRelated products of tetrahydrofuran

You may like
-
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
