(-)-Indolmycin
- CAS NO.:21200-24-8
- Empirical Formula: C14H15N3O2
- Molecular Weight: 257.29
- MDL number: MFCD01747966
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 14:59:12
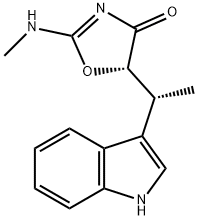
What is (-)-Indolmycin?
The Uses of (-)-Indolmycin
Indolmycin is a highly selective antibiotic which acts as a tryptophan anti-metabolite. Recent research has shown that indolmycin is active against Mycobacteria and H. pylori, and can stimulate transcription in Escherichia coli.
The Uses of (-)-Indolmycin
Indolmycin is an antibiotic that inhibits bacterial tryptophanyl-tRNA synthetase.
What are the applications of Application
Indolmycin is an antibiotic that inhibits bacterial tryptophanyl-tRNA synthetase
Definition
ChEBI: A member of the class of 1,3-oxazoles that is 1,3-oxazol-4(5H)-one which is substituted at the 2 and 5-pro-S positions by methylamino and [(1R)-1-(1H-indol-3-yl)ethyl] gro ps, respectively.
Biological Activity
indolmycin is an antibiotic.tryptophanyl-trna synthetase (trprs) catalyzes activation of tryptophan through atp and transfer to trnatrp , leading to translation of the genetic code for tryptophan.
in vitro
indolmycin was found to ba a bacteriostatic that showed good activity against methicillin-resistant s. aureus, methicillin-susceptible staphylococcus aureus, and vancomycin-intermediate s. aureus, including strains resistant to mupirocin or fusidic acid. spontaneous indolmycin-resistant mutants was observed at a lower frequency than those selected by mupirocin or fusidic acid and exhibited no cross-resistance with the comparative drugs. high-level resistance of indolmycin at its mic of 128 mg/l that was associated with an h43n mutation in tryptophanyl-trna synthetase, the target enzyme of indolmycin, led to loss of bacterial fitness. however, the locus responsible for low-level indolmycin resistance (indolmycin mics 8-32 mg/l) was not identified [1].
in vivo
animal study found that indolmycin could completely clear h. pylori in experimentally infected mongolian gerbils at a dose of 10 mg/kg. therefore, indolmycin could be regarded as a candidate for the treatment of h. pylori infection [2].
References
[1] hurdle jg, o'neill aj, chopra i. anti-staphylococcal activity of indolmycin, a potential topical agent for control of staphylococcal infections. j antimicrob chemother. 2004 aug;54(2):549-52. epub 2004 jul 8.
[2] vecchione jj, sello jk. a novel tryptophanyl-trna synthetase gene confers high-level resistance to indolmycin. antimicrob agents chemother 2009; 53: 3972-3980.
Properties of (-)-Indolmycin
| Melting point: | 209-210° |
| Boiling point: | 420.8±37.0 °C(Predicted) |
| alpha | D25 -214° (c = 2 in methanol) |
| Density | 1.36±0.1 g/cm3(Predicted) |
| storage temp. | Store at -20°C, protect from light |
| solubility | Soluble in ethanol;methanol;DMSO;dimethyl formamide |
| form | powder |
| pka | 16.65±0.30(Predicted) |
| color | white |
Safety information for (-)-Indolmycin
Computed Descriptors for (-)-Indolmycin
New Products
2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid 8-Bromo-3-methyl xanthine Maleic hydrazide o-Anisaldehyde 4-Bromobenzaldehyde 3-Fluorobenzaldehyde 2-Chlorobenzaldehyde 3-Amino-3-(3-fluorophenyl)propanoic acid 1-Propyl-4-piperidone 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2-Fluoro-6-iodobenzoic acid 3-Pyridineacrylic acid 1-Boc-4-cyanopiperidine ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate Vardenafil Bis-sulphonamide(Dimer) Nitroso Irbesartan N-Nitroso Nortriptyline Sulfonic acid Impurity 2-(p-Tolyl)acetaldehyde 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran

You may like
-
 Deuterated Styrene-d8 19361-62-7 97-99%View Details
Deuterated Styrene-d8 19361-62-7 97-99%View Details
19361-62-7 -
 1187989-89-4 97-99%View Details
1187989-89-4 97-99%View Details
1187989-89-4 -
 (S)-2-Bromobutanoic acid 97-99%View Details
(S)-2-Bromobutanoic acid 97-99%View Details
32659-49-7 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 methyl 3-amino-4-formylbenzoate 97-99%View Details
methyl 3-amino-4-formylbenzoate 97-99%View Details
212322-17-3 -
![3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%View Details
3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%View Details
1620885-59-7 -
![(13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) (13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%View Details
(13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%View Details
2590-41-2 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2
