Zipeprol dihydrochloride
- CAS NO.:34758-84-4
- Empirical Formula: C23H34Cl2N2O3
- Molecular Weight: 457.43
- MDL number: MFCD01748547
- EINECS: 252-192-2
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-15 10:43:21
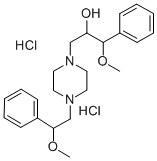
What is Zipeprol dihydrochloride?
Originator
Respilene,Winthrop,France,1973
The Uses of Zipeprol dihydrochloride
Zipeprol dihydrochloride is a piperazine ethanol derivative that is a widely used pediatric antitussive. Zipeprol dihydrochloride has cytotoxic and immunomodulative effects on human peripheral blood lymphocytes. Zipeprol dihydrochloride has a novel spectrum of action involving opioid and non-opioid effects with possible abuse potential.
Manufacturing Process
In a reactor provided with a mechanical stirrer, a reflux refrigerant and a
thermometer, there is introduced: 393 grams 1-[2-phenyl, 2-methoxy]ethyl
piperazine and 22 grams 3-phenyl-3-methoxy propylene oxide in 750 ml of
absolute ethanol.
When the slightly exothermic reaction (rise in temperature of about 20°C) has
ceased, heating is effected for 1.5 hours at 60°C. The product is then cooled
to 4°C and left to crystallize for about 12 hours. The precipitate is
centrifugated then recrystallized in 500 ml of absolute ethanol.
420 grams of the desired compound is thus obtained in the form of a white,
crystalline powder, melting point 83°C.
Therapeutic Function
Bronchodilator
Properties of Zipeprol dihydrochloride
| Melting point: | 231° |
Safety information for Zipeprol dihydrochloride
Computed Descriptors for Zipeprol dihydrochloride
New Products
Pentadecanoic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 2-Bromo-5-Iodopyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile Tetrahydropyranyl-4-acetic acid Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside 5-fluoro-1,3-benzodioxole 2-Bromo-6-fluoroaniline 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-oneRelated products of tetrahydrofuran



You may like
-
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
