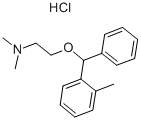
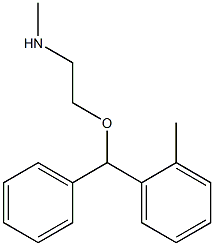
tofenacin hydrochloride
- CAS NO.:10488-36-5
- Empirical Formula: C17H21NO.ClH
- Molecular Weight: 291.82
- MDL number: MFCD00242926
- EINECS: 234-011-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-03-13 09:27:42
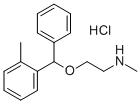
What is tofenacin hydrochloride?
Originator
Elamol,Brocades,UK,1971
The Uses of tofenacin hydrochloride
A metabolite of Orphenadrine (O695300). An antidepressant prodrug. Ondansetron USP Related Compound C.
Manufacturing Process
A mixture of 39.5 grams of 2-methylbenzhydrol, 200 ml of β-chloroethanol
and 10 ml of concentrated hydrochloric acid is boiled under reflux for 4 hours.
After cooling, the reaction mixture is poured into water and extracted with
petroleum ether (boiling range 40° to 60°C). The layers are separated and the
ethereal solution dried with sodium sulfate. It is then filtered. The filtrate is
concentrated by evaporation of the solvent. The residue is distilled under
reduced pressure to give 51.0 grams (yield 98%) of β-chloroethyl-2-
methylbenzhydryl ether, boiling at 156° to 158°C/2.5 mm.
A mixture of 51 grams of β-chloroethyl-2-methylbenzhydryl ether and 35
grams of methylamine in 140 ml of methanol is heated for 6 hours in a closed
vessel at a temperature of 125° to 135°C. After cooling, the reaction mixture
is poured into water and extracted with petroleum ether (boiling range 40° to
60°C). The ether layer is separated and washed with a 2 N hydrochloric acid
solution. The acidic layer is made alkaline and extracted with ether. The
ethereal solution is separated and dried with sodium sulfate. After filtration,
the solvent is evaporated and the residue distilled under reduced pressure.
There is thus obtained 40 grams (yield 80%) of N-methylaminoethyl-2-
methylbenzhydrylether boiling at 139° to 143°C/0.7 mm.
The base is dissolved in anhydrous ether, and an ethereal solution of
hydrochloric acid is added to form the hydrochloride of N-methylaminoethyl-2-
methylbenzhydryl ether. The salt is crystallized from a mixture of ethanol and
ether. Yield is 36 grams (78%); melting point 147° to 148°C.
brand name
Tofenacin is INN and BAN.
Therapeutic Function
Psychostimulant
Properties of tofenacin hydrochloride
| Melting point: | 147-148℃ (ethanol ethyl ether ) |
| solubility | Chloroform (Slightly), Methanol (Slightly), Water (Slightly) |
| form | Solid |
| color | White to Light Beige |
Safety information for tofenacin hydrochloride
Computed Descriptors for tofenacin hydrochloride
New Products
Methyl N-((2'-cyano-[1,1'-biphenyl]-4-yl)methyl)-N-nitroso-L-valinate Levofloxacin - Impurity B/ Levofloxacin Related Compound A Ivabradine R isomer Escitalopram - Impurity K Escitalopram Cyno dilol Impurity Paroxetine EP impurity I/Paroxetine Anhydrous EP Impurity I 2-Amino-5-Bromo-6-Chloropyridine 3-Amino-5-bromopicolinic acid 1-(7,8-Dinitro-4,5-dihydro-1H-1,5-methanobenzo[d]azepin-3(2H)-yl)-2,2,2-trifluoroethanone 2-Amino-3-Bromo-5-Cyanopyridine 2-Amino-3-Chloropyridine 2,5-Dibromo-3-MethylpyridineRelated products of tetrahydrofuran
![3-Methyl-7-phenyl-6-oxa-3-azabicyclo[6.4.0]dodeca-8,10,12-triene](https://img.chemicalbook.in/CAS/GIF/13669-70-0.gif)

You may like
-
 17471-10-2 Diphenhydramine EP impurity-A /Diphenhydramine Related Compound A NLT 95%View Details
17471-10-2 Diphenhydramine EP impurity-A /Diphenhydramine Related Compound A NLT 95%View Details
17471-10-2 -
 Dorzolamide Related Compound B/Dorzolamide EP Impurity B NLT 95%View Details
Dorzolamide Related Compound B/Dorzolamide EP Impurity B NLT 95%View Details
120279-90-5 -
 76824-16-3 Famotidine - Impurity D NLT 95%View Details
76824-16-3 Famotidine - Impurity D NLT 95%View Details
76824-16-3 -
 Metoprolol Impurity T NLT 95%View Details
Metoprolol Impurity T NLT 95%View Details
73723-85-0 -
 118-23-0 NLT 95%View Details
118-23-0 NLT 95%View Details
118-23-0 -
 Miconazole EP Impurity F/ Miconazole USP Related Compound F 47447-55-2 NLT 95%View Details
Miconazole EP Impurity F/ Miconazole USP Related Compound F 47447-55-2 NLT 95%View Details
47447-55-2 -
 2407630-84-4 NLT 95%View Details
2407630-84-4 NLT 95%View Details
2407630-84-4 -
 573704-41-3 NLT 95%View Details
573704-41-3 NLT 95%View Details
573704-41-3
