Sodium hydroxide
Synonym(s):NaOH;Sodium hydroxide;Caustic Soda;Sodium hydroxide solution;Sodium Hydrate
- CAS NO.:1310-73-2
- Empirical Formula: NaOH
- Molecular Weight: 39.99711
- MDL number: MFCD00011405
- EINECS: 215-185-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-12 09:17:10
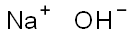
What is Sodium hydroxide?
Absorption
There are no quantitative data for the absorption of sodium hydroxide through the skin. Solutions which contain 50 % sodium hydroxide have been shown to be corrosive and lethal when applied dermally to mice .
Toxicity
Human poisoning cases indicate that a dose of 10 grams orally is fatal . Sodium hydroxide is toxic by oral ingestion .Sodium hydroxide is corrosive to all tissues. Concentrated vapors lead to serious damage to the eyes and respiratory system. Oral ingestion of sodium hydroxide, which occurs frequently in children, causes severe tissue necrosis, with stricture formation of the esophagus, often resulting in death. Contact with the skin may result in contact dermatitis, hair loss, as well as necrosis due to severe irritation.
Description
Sodium hydroxide, also known as lye and caustic soda, is a highly caustic substance that is used used in small amounts in cosmetics to establish and hold the pH of a product.Sodium Hydroxide is a extremely important compound in our lives because it has so many uses. It is a very common base used in the chemical industry and is used for many things, many of which occur in our daily lives.
References
https://pubchem.ncbi.nlm.nih.gov/compound/sodium_hydroxide
Chemical properties
Sodium hydroxide occurs as a white or nearly white fused mass. It is available in small pellets, flakes, sticks, and other shapes or forms. It is hard and brittle and shows a crystalline fracture. Sodium hydroxide is very deliquescent and on exposure to air it rapidly absorbs carbon dioxide and water.
Physical properties
White orthorhombic crystals, produced in the form of pellets, lumps, sticks, beads, chips, flakes or solutions; hygroscopic; very corrosive; rapidly absorbs CO2 and water from the air; density 2.13 g/cm3; melts at 323°C; vaporizes at 1388°C; vapor pressure 1 torr at 739°C and 5 torr at 843°C; very soluble in water (110 g/100mL at room temperature), generating heat on dissolution; aqueous solutions highly alkaline, pH of 0.5% solution about 13 and 0.05% solution about 12; soluble in methanol, ethanol and glycerol (23.8 g/100 mL methanol and 13.9 g/100 mL ethanol at ambient temperatures.).
The Uses of Sodium hydroxide
Sodium hydroxide is one of the most important industrial chemicals. It is used in manufacturing a large number of compounds including several sodium salts, in treating cellulose for producing rayon and cellophane, and in manufacturing soaps, detergents, pulp, and paper. Sodium hydroxide is a common neutralizing agent for acids in acid-base titrations and petroleum refining. Another major application is extracting metals from their ores where alkali fusion, such as fusion with caustic soda, often is applied to open the ores. Additionally, sodium hydroxide is used to precipitate metals as hydroxides. Other uses are in reclaiming rubber, dissolving casein in plastics production, refining vegetable oils, processing textiles, as an eluant in ion chromatography, etching and electroplating, and as a laboratory reagent. Sodium hydroxide also is used as a strong base in many organic synthesis and base-catalyzed reactions. Sodium Hydroxide is a strong base and acid neutralizer also used in the staining procedure of cells.
Background
Sodium hydroxide is also known as lye or soda , or caustic soda . At room temperature, sodium hydroxide is a white crystalline odorless solid that absorbs moisture from the air. It is a synthetically manufactured substance. When dissolved in water or neutralized with acid it releases substantial amounts of heat, which may prove sufficient to ignite combustible materials. Sodium hydroxide is highly corrosive .
Indications
Used to destroy or kill the nail matrix (matrixectomies) .
Preparation
Sodium hydroxide is manufactured together with chlorine by electrolysis of sodium chloride solution. Various types of electrolytic cells are used commercially. They include the mercury cell, the diaphragm cell, and the membrane cell.
A saturated solution of brine is electrolyzed. Chlorine gas is liberated at the anode and sodium ion at the cathode. Decomposition of water produces hydrogen and hydroxide ions. The hydroxide ion combines with sodium ion forming NaOH. The overall electrolytic reactions may be represented as:
2Na+ + 2Cl-+ 2H2O → Cl2 (g) + H2 (g) + 2NaOH (aq)
The mercury cell proceeds in two stages that occur separately in two cells. The first is known as the brine cell or the primary electrolyzer in which sodium ion deposits on the mercury cathode forming amalgam, while chlorine gas is liberated at the anode:
Na+ + Cl–→ Na-Hg (cathode) + ½Cl2(g) (anode)
In the second cell, known as the decomposer cell, a graphite cathode is used while sodium amalgam serves as the anode. Water reacts with the sodium metal of the amalgam in the decomposer:
Na-Hg + H2O → Na+ + OH– + ½H2↑ + Hg
In chlor-alkali diaphragm cells, a diaphragm is employed to separate chlorine liberated at the anode from the sodium hydroxide and hydrogen generated at the cathode. Without a diaphragm, the sodium hydroxide formed will combine with chlorine to form sodium hypochlorite and chlorate. In many cells, asbestos diaphragms are used for such separation. Many types of diaphragm cells are available.
Sodium hydroxide is produced either as an anhydrous solid or as a 50% aqueous solution.
Definition
The most important commercial caustic.
Production Methods
Sodium hydroxide is manufactured by electrolysis of brine using
inert electrodes. Chlorine is evolved as a gas at the anode and
hydrogen is evolved as a gas at the cathode. The removal of chloride
and hydrogen ions leaves sodium and hydroxide ions in solution.
The solution is dried to produce the solid sodium hydroxide.
A second method uses the Kellner–Solvay cell. Saturated sodium
chloride solution is electrolyzed between a carbon anode and a
flowing mercury cathode. In this case the sodium is produced at the
cathode rather than the hydrogen because of the readiness of
sodium to dissolve in the mercury. The sodium–mercury amalgam is
then exposed to water and a sodium hydroxide solution is
produced.
Reactions
Sodium hydroxide is strongly alkaline and can react with acids to form salts and water.
Sodium hydroxide reacts with acidic oxides to form salt and water, so sodium hydroxide can be used to absorb acid gases in the laboratory or industrially.
Sodium hydroxide can react with aqueous solutions of many metal salts to form sodium salts and metal hydroxides When sodium hydroxide and ammonia salt are heated together, it can release ammonia.
Sodium hydroxide is highly corrosive, so that the glass bottles storing sodium hydroxide solutions must be rubber stoppers, and glass stoppers should not be used to prevent a chemical reaction from opening.
Sodium hydroxide is an important industrial raw material, and can be produced by electrolysis of saline solution industrially.
Air & Water Reactions
Soluble in water. Dissolution can liberate enough heat to cause steaming and spattering and ignite adjacent combustible material [Haz. Chem. Data 1966].
Reactivity Profile
CAUSTIC SODA (Sodium hydroxide) is a strong base. Reacts rapidly and exothermically with acids, both organic and inorganic. Readily absorbs moisture from the air to give caustic semi-solids that attack aluminum and zinc with the evolution of flammable hydrogen gas. Catalyzes the polymerization of acetaldehyde and other polymerizable compounds; these reactions can occur violently, for example, acrolein polymerizes with extreme violence when put in contact with alkaline materials such as sodium hydroxide. Reacts with great violence with phosphorus pentaoxide when initiated by local heating [Mellor 8 Supp.3:406 1971]. Contact (as a drying agent) with tetrahydrofuran, which often contains peroxides, may be hazardous---explosions have occurred in such a use of the chemically similar potassium hydroxide [NSC Newsletter Chem. Soc. 1967].
Hazard
Corrosive to tissue in presence of mois- ture, strong irritant to tissue (eyes, skin, mucous membranes, and upper respiratory tract), poison by ingestion.
Health Hazard
Sodium hydroxide is a highly corrosive substancethat causes damage to human tissues.Its action on the skin is somewhat differentfrom acid burns. There is no immediate pain,but it penetrates the skin. It does not coagulateprotein to prevent its further penetration,and thus the caustic burn can become severeand slow healing. Spilling of its concentratedsolutions into the eyes can result in severeirritation or permanent injury.
It is toxic by ingestion as well as inhalationof its dust. Although the oral toxicity ofa 5–10% solution of caustic soda was foundto be low in test animals, high dosages atgreater concentrations can cause vomiting,prostration, and collapse. The oral lethal dosein rabbits is 500 mg/kg (NIOSH 1986).
Sodium hydroxide dusts or aerosols areirritating to the eyes, nose, and throat. Prolongedexposure to high concentrations in airmay produce ulceration of the nasal passage.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated.
Flammability and Explosibility
Sodium hydroxide and potassium hydroxide are not flammable as solids or aqueous solutions.
Pharmaceutical Applications
Sodium hydroxide is widely used in pharmaceutical formulations to adjust the pH of solutions. It can also be used to react with weak acids to form salts.
Industrial uses
Caustic soda (NaOH) is regarded as the strongest alkaline pH regulator. Caustic soda
is a very active substance and is highly corrosive. The bulk of caustic soda is manufactured
by electrolysis of saturated brines (NaCl). Caustic soda has a very strong pHregulating
capability (i.e. from pH 7 to pH 14) at a relatively low dosage compared to
other alkaline substances. Commercially, caustic soda is available in anhydrous form,
but in most mining applications the caustic soda is supplied as a 50% solution.
In the mineral processing industry, sodium hydroxide is mostly used for alkalinity control
during the processing of non-metallic minerals. In base metal flotation, the use of
sodium hydroxide is rare.
Pharmacokinetics
Sodium Hydroxide 10% forms a strongly alkaline and caustic solution. As a caustic agent, it is used to destroy organic tissue by chemical action .
Safety
Sodium hydroxide is widely used in the pharmaceutical and food
industries and is generally regarded as a nontoxic material at low
concentrations. At high concentrations it is a corrosive irritant to
the skin, eyes, and mucous membranes.
LD50 (mouse, IP): 0.04 g/kg
LD50 (rabbit, oral): 0.5 g/kg
Potential Exposure
NaOH is utilized to neutralize acids and make sodium salts in petroleum refining, viscose rayon; cellophane, plastic production; and in the reclamation of solutions of their salts. It is used in the manufacture of mercerized cotton, paper, explosives, and dyestuffs in metal cleaning; electrolytic extraction of zinc; tin plating; oxide coating; laundering, bleaching, dishwashing; and it is used in the chemical industries.
Sodium hydroxide application
Caustic soda is one of the most widely usedchemicals. It is used to neutralize acids; tomake sodium salts; to precipitate metals astheir hydroxides; in petroleum refining; in thesaponification of esters; in the treatment ofcellulose, plastics, and rubber; and in numeroussynthetic and analytical applications.
Storage
Sodium hydroxide should be stored in an airtight nonmetallic container in a cool, dry place. When exposed to air, sodium hydroxide rapidly absorbs moisture and liquefies, but subsequently becomes solid again owing to absorption of carbon dioxide and formation of sodium carbonate.
Shipping
UN1823 NaOH, solid, Hazard class: 8; Labels: 8-Corrosive material. UN1824 NaOH, solution, Hazard class: 8; Labels: 8-Corrosive material
Incompatibilities
Sodium hydroxide is a strong base and is incompatible with any compound that readily undergoes hydrolysis or oxidation. It will react with acids, esters, and ethers, especially in aqueous solution.
Waste Disposal
Discharge into tank containing water, neutralize, then flush to sewer with water.
Regulatory Status
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (dental preparations; injections; inhalations; nasal, ophthalmic, oral, otic, rectal, topical, and vaginal preparations). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Properties of Sodium hydroxide
| Melting point: | 681 °C(lit.) |
| Boiling point: | 1390°C |
| Density | 1.515 g/mL at 20 °C |
| vapor density | <1 (vs air) |
| vapor pressure | 1 mm Hg ( 745 °C) |
| refractive index | 1,473-1,475 |
| Flash point: | 176-178°C |
| storage temp. | room temp |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| form | beads |
| appearance | White solid |
| color | White |
| Specific Gravity | 2.13 |
| PH Range | 13 - 14 |
| Odor | Odorless |
| PH | 10.98(1 mM solution);11.95(10 mM solution);12.88(100 mM solution); |
| Water Solubility | SOLUBLE |
| Decomposition | 176-178 ºC |
| Sensitive | Air Sensitive & Hygroscopic |
| λmax | λ: 260 nm Amax: 0.015 λ: 280 nm Amax: 0.01 |
| Merck | 14,8627 |
| Exposure limits | TLV-TWA air 2 mg/m3 (OSHA); ceiling
2 mg/m3 (ACGIH) and 2 mg/m3/15 min
(NIOSH). |
| Dielectric constant | 57.5(25℃) |
| Stability: | hygroscopic |
| CAS DataBase Reference | 1310-73-2(CAS DataBase Reference) |
| NIST Chemistry Reference | Sodium hydroxide(1310-73-2) |
| EPA Substance Registry System | Sodium hydroxide (1310-73-2) |
Safety information for Sodium hydroxide
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05 |
| GHS Hazard Statements |
H290:Corrosive to Metals H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P234:Keep only in original container. P260:Do not breathe dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Sodium hydroxide
Sodium hydroxide manufacturer
CEFA CILINAS BIOTICS PVT LTD
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran

You may like
-
 Caustic Soda Flakes 98%View Details
Caustic Soda Flakes 98%View Details -
 Caustic Soda flakes 99%View Details
Caustic Soda flakes 99%View Details -
 Caustic Soda Flakes 99%View Details
Caustic Soda Flakes 99%View Details -
 Caustic Soda Flakes 99%View Details
Caustic Soda Flakes 99%View Details -
 Sodium hydroxide 99%View Details
Sodium hydroxide 99%View Details -
 Sodium hydroxide 98%View Details
Sodium hydroxide 98%View Details -
 Sodium hydroxide 98%View Details
Sodium hydroxide 98%View Details -
 Sodium Hydroxide CASView Details
Sodium Hydroxide CASView Details
