SEDANOLIDE
- CAS NO.:6415-59-4
- Empirical Formula: C12H18O2
- Molecular Weight: 194.27
- MDL number: MFCD00171327
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-22 18:57:36
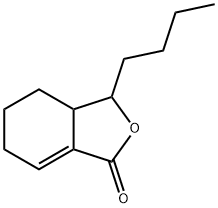
What is SEDANOLIDE?
The Uses of SEDANOLIDE
Sedanolide is a natural phthalide isolates from seed oil of Umbelliferae family. It induces the expression of glutathione S-transferase and reduces chemical induced carcinogenesis in mice.
What are the applications of Application
Sedanolide is an inducer of GST and inhibitor of Cox-1, Cox-2, Topo I, Topo II, and carcinogenesis
Definition
ChEBI: Sedanolide is a member of 2-benzofurans.
Biological Activity
sedanolide is a natural compound produced in edible umbelliferous plants, such as celery seed oil [1].in hepg2 and caco-2 cells, treatment with sedanolide (7-500 μm) for 24h showed no effect on cell viability. in hepg2 cells cultured in sedanolide-free medium, sedanolide (500 μm) treatment for 72h decreased cell viability. pretreatment with sedanolide (100 μm) for 24 h and exposement to either h2o2 or tbooh did not exhibit statistically significant difference in viability from controls. in hepg2 following 24-h incubation with 500 μm sedanolide, a significant increase in dna strand breaks was observed. sedanolide did not modulate h2o2- and tbooh-induced dna damage. sedanolide was relatively nontoxic to cells in culture [1]. sedanolide (sn) possesses antioxidant effects. in human liver cancer (j5) cells, treatment with sedanolide suppressed j5 cell viability by inducing autophagy. sedanolide decreased protein expression levels of phosphoinositide 3-kinase (pi3k)-i, mammalian target of rapamycin (mtor) and akt and increased pi3k-iii, lc3-ii and beclin-1 protein levels. sedanolide increased the cytosolic phosphorylation of inhibitor of kappa b (iκb) and nuclear p65 and the dna-binding activity of nf-κb. sedanolide induced j5 cell autophagy by regulating pi3k, p53 and nf-κb autophagy-associated signaling pathways in j5 cells [2]. sedanolide (100 μg/ml) inhibited cyclooxygenases-1 and -2 at 250 pg/ml and blocked topoisomerase-i and-ii activity [3].
References
[1] woods j a, jewell c, o'brien n m. sedanolide, a natural phthalide from celery seed oil: effect on hydrogen peroxide and tert-butyl hydroperoxide-induced toxicity in hepg2 and caco-2 human cell lines[j]. in vitro & molecular toxicology: a journal of basic and applied research, 2001, 14(3): 233-240.
[2] hsieh s l, chen c t, wang j j, et al. sedanolide induces autophagy through the pi3k, p53 and nf-κb signaling pathways in human liver cancer cells[j]. international journal of oncology, 2015, 47(6): 2240-2246
[3] momin r a, nair m g. antioxidant, cyclooxygenase and topoisomerase inhibitory compounds from apium graveolens linn. seeds[j]. phytomedicine, 2002, 9(4): 312-318.
Properties of SEDANOLIDE
| Melting point: | 30~31℃ |
| Boiling point: | 342.0±11.0 °C(Predicted) |
| Density | 1.03 |
| storage temp. | Store at -20°C, protect from light |
| solubility | insoluble in H2O; ≥52.8 mg/mL in EtOH; ≥6.65 mg/mL in DMSO |
| form | White to off-white solid. |
| color | White to off-white |
| Odor | at 100.00 %. herbal celery |
| CAS DataBase Reference | 6415-59-4(CAS DataBase Reference) |
Safety information for SEDANOLIDE
Computed Descriptors for SEDANOLIDE
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloropyridazine 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 6-Methyl-2-pyridinemethanol 2-Pyridinecarboxaldehdye tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 1-Indanone 2-Chloromethyl-6-methyl-pyridine 4-Morpholinoaniline Abiraterone Ethyl Ether 1-nitro-3,5-dimethyl adamantine (NMEM) Vardenafil Bis-sulphonamide(Dimer) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran
![2-THIO(3-IODOBENZYL)-5-(1-PYRIDYL)-[1,3,4]-OXADIAZOLE](https://img.chemicalbook.in/StructureFile/ChemBookStructure7/GIF/CB2201711.gif)
![S-[N-(3-PHENYLPROPYL)(THIOCARBAMOYL)]-L-CYSTEINE](https://img.chemicalbook.in/CAS/GIF/137915-13-0.gif)
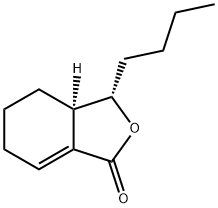
You may like
-
![3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
2304754-51-4 -
 1126-74-5 99%+View Details
1126-74-5 99%+View Details
1126-74-5 -
 2-Fluoro-6-iodobenzoic acid 99%+View Details
2-Fluoro-6-iodobenzoic acid 99%+View Details
111771-08-5 -
 120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 -
 2-pyridineacetonitrile 2739-97-1 99%+View Details
2-pyridineacetonitrile 2739-97-1 99%+View Details
2739-97-1 -
 4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
138163-08-3 -
![ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
313544-31-9 -
 1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
91419-52-2
