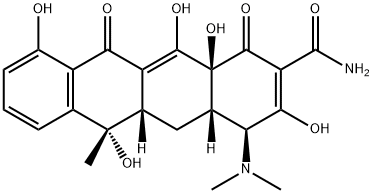
ROLITETRACYCLINE
- CAS NO.:751-97-3
- Empirical Formula: C27H33N3O8
- Molecular Weight: 527.57
- MDL number: MFCD00864995
- EINECS: 212-031-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-06-08 09:02:53
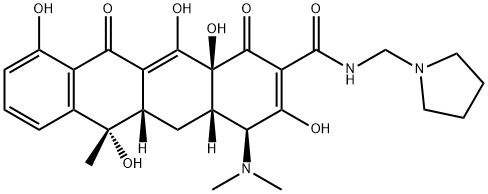
What is ROLITETRACYCLINE?
Toxicity
Symptoms of overdose include anorexia, nausea, diarrhoea, glossitis, dysphagia, enterocolitis and inflammatory lesions (with monilial overgrowth) in the anogenital region, skin reactions such as maculopapular and erythematous rashes, exfoliative dermatitis, photosensitivity, hypersensitivity reactions such as urticaria, angioneurotic oedema, anaphylaxis, anaphyl-actoid purpura, pericarditis, and exacerbation of systemic lupus erythematosus, benign intracranial hypertension in adults disappearing on discontinuation of the medicine, haematologic abnormalities such as haemolytic anaemia, thrombocytopenia, neutropenia, and eosinophilia. LD50=262 mg/kg (I.P. in rat).
Description
Rolitetracycline was synthesized in 1958. Bristol-Myers Laboratories prepared it from tetracycline by introducing a pyrrolidinylmethyl moiety. This antibiotic is very soluble in water and more stable than tetracycline under acidic conditions. Rolitetracycline is used by intravenous injection, and its nitrate is used by both intravenous and intramuscular injections for therapy of the same infections as those treated by tetracycline.
The Uses of ROLITETRACYCLINE
Rolitetracycline is a tetracycline antibiotic that may be given intravenously or intramuscularly in serious bacterial infections when oral administration is not practicable.
The Uses of ROLITETRACYCLINE
neuromuscular blocker
The Uses of ROLITETRACYCLINE
Rolitetracycline, launched in the late 1950s, was the first of the semi-synthetic tetracyclines. Rolitetracycline is formed by a Mannich condensation of formaldehyde and pyrrolidine with tetracycline. Rolitetracycline is a pro-drug of tetracycline, in which the pyrrolidine moiety improves bioavailability compared with tetracycline. Rolitetracycline has broad spectrum Gram positive activity in vivo, but pH instability limits use to parenteral administration. The intrinsic in vitro activity and SARs for this region of the tetracycline molecule have not been extensively investigated.
Indications
Rolitetracycline is a broad-spectrum antibiotic used in cases needing high concentrations or when oral administration is impractical.
Background
A pyrrolidinylmethyl tetracycline.
What are the applications of Application
Rolitetracycline is a tetracycline antibiotic
Definition
ChEBI: A derivative of tetracycline in which the amide function is substituted with a pyrrolidinomethyl group.
brand name
Syntetrin (Bristol-Myers Squibb).
General Description
Rolitetracycline, N-(pyrrolidinomethyl)tetracycline(Syntetrin), was introduced for use by intramuscular or intravenousinjection. This derivative is made by condensingtetracycline with pyrrolidine and formaldehyde in the presenceof tert-butyl alcohol. It is very soluble in water (1 g dissolvesin about 1 mL) and provides a means of injecting theantibiotic in a small volume of solution. It has been recommendedfor cases when the oral dosage forms are not suitable,but it is no longer widely used.
Pharmaceutical Applications
2-N-pyrrolidinomethyl-tetracycline. A semisynthetic derivative
of tetracycline supplied as the nitrate sesquihydrate for
parenteral use.
It is not absorbed from the gastrointestinal tract. It is highly
soluble and therefore can be administered parenterally. Peak
plasma concentrations of 4–6 mg/L occur at 0.5–1 h after
350 mg intravenously. The plasma elimination half-life is 5–8 h.
About 50% of the dose is excreted in the urine, producing
high concentrations.
Intravenous administration is occasionally accompanied by
abnormal taste, shivering and rigors, hot flushes, facial reddening,
dizziness and, rarely, circulatory collapse. Symptoms
of myasthenia gravis have occasionally been exacerbated.
Metabolism
Not Available
Properties of ROLITETRACYCLINE
| Melting point: | 163.5°C |
| Boiling point: | 608.16°C (rough estimate) |
| Density | 1.2902 (rough estimate) |
| refractive index | 1.7120 (estimate) |
| storage temp. | -20°C Freezer, Under inert atmosphere |
| solubility | DMSO (Slightly), Methanol (Very Slightly) |
| form | Solid |
| pka | pKa 7.4 (Uncertain) |
| color | Very Dark Red to Very Dark Brown |
| Water Solubility | >20g/L(21 ºC) |
Safety information for ROLITETRACYCLINE
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for ROLITETRACYCLINE
New Products
Dexamethasone Sodium Phosphate p-Anisaldehyde m-Tolualdehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid Raloxifene EP Impurity D N-Nitroso Nortriptyline Nitroso Irbesartan 2-(p-Tolyl)acetaldehyde Sulfonic acid Impurity 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 5-Chloro-2-Methoxy-4-Picoline 2-Amino-6-nitrobenzoic Acid (6-Nitroanthranilic acid) Cis-3,5 Diacetoxy-1-cyclopentene 3-Bromo-2-Methoxy-4-Methylpyridine 2-Amino-5-Chloro-3-Nitro-6-picoline 3-Methoxy-5-methylpyrazin-2-amine Pentachlorobenzonitrile Budesonide Nicotinic acid RAPAMYCIN Alpha CyclodextrinRelated products of tetrahydrofuran

You may like
-
 88660-53-1 >99%View Details
88660-53-1 >99%View Details
88660-53-1 -
 12029-98-0 >98%View Details
12029-98-0 >98%View Details
12029-98-0 -
 1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 -
 Hydrocortisone 50-23-7 >98%View Details
Hydrocortisone 50-23-7 >98%View Details
50-23-7 -
 106-43-4 Parachlorotoluene >99%View Details
106-43-4 Parachlorotoluene >99%View Details
106-43-4 -
 Pyrrolidin >98%View Details
Pyrrolidin >98%View Details
123-75-1 -
 cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
7212-40-0 -
 Para Chloro toluene (PCT) 106-43-4 >99%View Details
Para Chloro toluene (PCT) 106-43-4 >99%View Details
106-43-4
