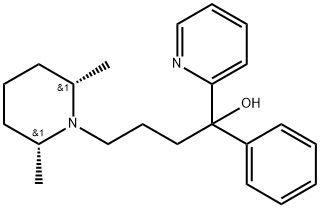
Pirmenol hydrochloride
- CAS NO.:61477-94-9
- Empirical Formula: C22H31ClN2O
- Molecular Weight: 0
- MDL number: MFCD01691662
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-03-13 09:27:42
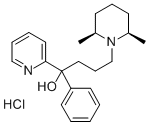
What is Pirmenol hydrochloride?
Description
Pirrnenol hydrochloride is a long-lasting Class la antiarrhythmic agent introduced in Japan in 1994. The spectrum of antiarrhythmic activity of pirmenol includes atrial and ventricular arrhythmias of diverse etiology. In clinical trials, pirmenol was found effective in the short- and mid-term treatment of chronic premature ventricular beats, with or without underlying heart disease. In patients with supraventricular tachycardia, a single oral dose of pirmenol was highly effective in suppressing induction of tachycardia with adequate hemodynamic tolerance. Since pirmenol is highly effective by both intravenous and oral routes, it enables immediate suppression, prevention, or termination of cardiac arrhythmias and facilitates the transition to oral prophylactic therapy.
Originator
Warner-Lamber (U.S.A.)
The Uses of Pirmenol hydrochloride
Cardiac depressant (anti-arrhythmic).
brand name
Pirmavar (Parke-Davis);Plmenol.
Properties of Pirmenol hydrochloride
| Melting point: | 171-172 °C |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | DMSO : ≥ 28 mg/mL (74.68 mM) |
| form | Solid |
| color | White to yellow |
Safety information for Pirmenol hydrochloride
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Pirmenol hydrochloride
New Products
2-(2-aminoethyl) benzenesulfonamide Gemcitabine 3-benzoate impurity Dihydroxyphenylacetone Ornidazole EP impurity D Acetyl amino diol impurity Valsartan Benzyl Ester Impurity B N,N'-Dicyclohexylcarbodiimide 3-Bromo-2-Methyl-5-Nitropyridine 5-Bromo-3-Methyl-2-Pyridinecarboxylic acid 2-Amino-4-Methylpyridine 4-Aminopyridine 4-dimethylaminopyridineRelated products of tetrahydrofuran

You may like
-
 4-(Methylnitrosoamino)-butanal NLT 95%View Details
4-(Methylnitrosoamino)-butanal NLT 95%View Details
339362-86-6 -
 848696-99-1 NLT 95%View Details
848696-99-1 NLT 95%View Details
848696-99-1 -
 Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
1384752-15-1 -
 615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 -
 175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 -
 S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
1346605-15-9 -
 Valsartan Related Compound A NLT 95%View Details
Valsartan Related Compound A NLT 95%View Details
137862-87-4 -
 Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
124750-49-8
