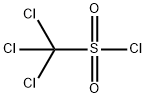
Perchloromethylmercaptan
Synonym(s):Perchloromethyl mercaptan
- CAS NO.:594-42-3
- Empirical Formula: CCl4S
- Molecular Weight: 185.89
- MDL number: MFCD00037054
- EINECS: 209-840-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-10 10:27:26
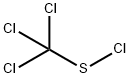
What is Perchloromethylmercaptan?
Chemical properties
CLEAR YELLOW LIQUID
Chemical properties
Perchloromethyl mercaptan is a pale yellow oily liquid with a foul-smelling, unbearable, acrid odor
The Uses of Perchloromethylmercaptan
Organic synthesis, dye intermediate, fumigant.
The Uses of Perchloromethylmercaptan
Production of fungicides; vulcanizing accelerator in rubber industry
The Uses of Perchloromethylmercaptan
Perchloromethyl Mercaptan is used as a reactant in the preparation of thio- and selenoesters of triflic acid via a route to thio- and selenosulfonates from disulfides and diselenides.
Synthesis Reference(s)
Tetrahedron, 48, p. 8065, 1992 DOI: 10.1016/S0040-4020(01)80477-4
General Description
A yellow oily liquid with an offensive odor. Insoluble in water. Density 1.72 g / cm3. Hence sinks in water. Nonflammable but supports combustion. Very toxic by inhalation or skin absorption.
Air & Water Reactions
Insoluble in water. Slowly decomposed by moisture in the air. Reacts with hot water to give carbon dioxide, hydrochloric acid and sulfur.
Reactivity Profile
Perchloromethylmercaptan is incompatible with acids, diazo and azo compounds, halocarbons, isocyanates, aldehydes, alkali metals, nitrides, hydrides, and other strong reducing agents. Reactions with these materials generate heat and in many cases hydrogen gas. Reacts readily with oxidizing agents.
Health Hazard
May cause death or permanent injury after short exposure to small quantities, strong irritant to eyes and skin. Inhalation may cause severe irritation of the upper respiratory tract. It also is a strong irritant to the eyes and skin. Brief exposure to lower concentrations may produce central nervous system depression and lung, liver, and heart congestion. Severe exposures may be fatal. Exposure of eyes may lead to severe conjunctivitis or corneal damage. The liquid is irritating to the skin, and may be absorbed through the skin in quantities sufficient to cause general toxic effects. Ingestion may cause damage to mucous membranes and result in pain and burning of the mouth and throat, nausea, vomiting, cramps, and diarrhea. In severe cases, tissue ulceration and CNS depression may occur.
Fire Hazard
Very irritating vapors formed from hot material; may form phosgene gas, hydrogen chloride, and sulfur dioxide. At high temperatures Perchloromethylmercaptan will decompose to carbon tetrachloride, sulfur chloride, heavy oil polymers, phosgene gas, hydrogen chloride, and sulfur dioxide. Reacts with iron or steel, evolving carbon tetrachloride. Corrosive to most metals. Reacts with water only when hot to give carbon dioxide, hydrochloric acid, and sulfur. Hazardous polymerization may not occur.
Safety Profile
Poison by ingestion, inhalation, and intravenous routes. A severe skin, eye, and mucous membrane irritant. When heated to decomposition it emits very toxic fumes of Cland SOx. See also MERCAPTANS
Potential Exposure
Perchloromethyl mercaptan is used as an intermediate for the synthesis of dyes and fungicides, such as Captan and Folpet. This chemical has been considered as a warfare tear gas because of its highly irritant properties.
Shipping
UN1670 Perchloromethyl mercaptan, Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Inhalation Hazard Zone B.
Incompatibilities
Water contact forms HCl, sulfur and carbon dioxide. Reacts with alkalies, amines, hot water; alcohols, oxidizers, reducing agents; iron, and steel. Attacks most metals.
Waste Disposal
Incineration together with a flammable solvent in a furnace equipped with afterburner and scrubber.
Properties of Perchloromethylmercaptan
| Boiling point: | 146-148 °C(lit.) |
| Density | 1.7 g/mL at 25 °C(lit.) |
| vapor density | 6.41 (vs air) |
| vapor pressure | 2.02 psi ( 20 °C) |
| refractive index | n |
| storage temp. | -20°C Freezer |
| solubility | Chloroform, Ethyl Acetate |
| form | Liquid |
| color | Clear yellow |
| CAS DataBase Reference | 594-42-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Methanesulfenyl chloride, trichloro-(594-42-3) |
| EPA Substance Registry System | Perchloromethyl mercaptan (594-42-3) |
Safety information for Perchloromethylmercaptan
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Skull and Crossbones Acute Toxicity GHS06 |
| GHS Hazard Statements |
H301:Acute toxicity,oral H312:Acute toxicity,dermal H314:Skin corrosion/irritation H330:Acute toxicity,inhalation |
| Precautionary Statement Codes |
P260:Do not breathe dust/fume/gas/mist/vapours/spray. P270:Do not eat, drink or smoke when using this product. P280:Wear protective gloves/protective clothing/eye protection/face protection. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Perchloromethylmercaptan
New Products
Dexamethasone Sodium Phosphate p-Anisaldehyde m-Tolualdehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid Raloxifene EP Impurity D N-Nitroso Nortriptyline Nitroso Irbesartan 2-(p-Tolyl)acetaldehyde Sulfonic acid Impurity 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 5-Chloro-2-Methoxy-4-Picoline 2-Amino-6-nitrobenzoic Acid (6-Nitroanthranilic acid) Cis-3,5 Diacetoxy-1-cyclopentene 3-Bromo-2-Methoxy-4-Methylpyridine 2-Amino-5-Chloro-3-Nitro-6-picoline 3-Methoxy-5-methylpyrazin-2-amine Pentachlorobenzonitrile Budesonide Nicotinic acid RAPAMYCIN Alpha CyclodextrinRelated products of tetrahydrofuran



You may like
-
 Perchloromethyl mercaptan, 97% CAS 594-42-3View Details
Perchloromethyl mercaptan, 97% CAS 594-42-3View Details
594-42-3 -
 Perchloromethyl mercaptan 98% CAS 594-42-3View Details
Perchloromethyl mercaptan 98% CAS 594-42-3View Details
594-42-3 -
 12029-98-0 >98%View Details
12029-98-0 >98%View Details
12029-98-0 -
 Hydrocortisone 50-23-7 >98%View Details
Hydrocortisone 50-23-7 >98%View Details
50-23-7 -
 106-43-4 Parachlorotoluene >99%View Details
106-43-4 Parachlorotoluene >99%View Details
106-43-4 -
 Pyrrolidin >98%View Details
Pyrrolidin >98%View Details
123-75-1 -
 cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
7212-40-0 -
 Para Chloro toluene (PCT) 106-43-4 >99%View Details
Para Chloro toluene (PCT) 106-43-4 >99%View Details
106-43-4
