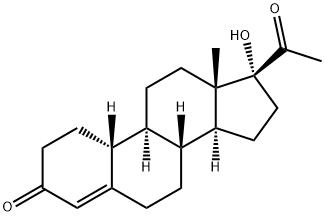
GESTONORONE CAPROATE
- CAS NO.:1253-28-7
- Empirical Formula: C26H38O4
- Molecular Weight: 414.58
- MDL number: MFCD00867860
- EINECS: 215-010-2
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-21 10:31:43
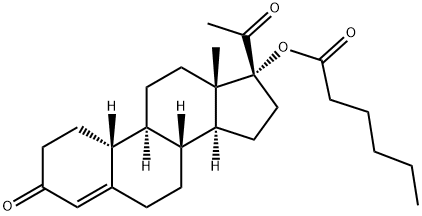
What is GESTONORONE CAPROATE?
Originator
Depostat,Schering AG
The Uses of GESTONORONE CAPROATE
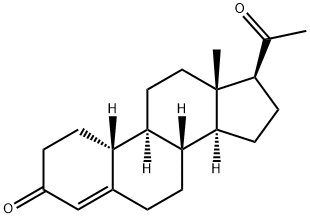
Gestonorone Caproate, is a pro-drug of Gestonorone, which is a steroidal progestin. It has been shown to have accelerated body weight gain and caused the atrophy of the prostate, uterus, and seminal vesicles in rats. It is also an inhibitor of the reductive pathway of Testosterone (T155000) metabolism.
The Uses of GESTONORONE CAPROATE
Gestonorone Capronate, is a pro-drug of Gestonorone, which is a steroidal progestin. It has been shown to have accelerated body weight gain and caused the atrophy of the prostate, uterus, and seminal vesicles in rats. It is also an inhibitor of the reductive pathway of Testosterone (T155000) metabolism.
Definition
ChEBI: Gestonorone caproate is an organic molecular entity.
Manufacturing Process
2 Methods of producing of 17-α-hydroxyl-19-norprogesteron-17-capronate:
1. To a solution of 1.0 g 17-α-hydroxy-19-norprogesteron in 32 ml capronic
acid anhydride 1.32 g p-toluesulfonate (1 mole hydrate) were added, and
allowed to stand for 3 h at 37°C. To the solution 1.43 ml conc. hydrochloric
acid in 143 ml methanol were added and all this also for 1 h was left under
N2. Then mixture was washed with water and treated with ether. Ether extract
was washed with water, and dried with Na2SO4. After that ether was distilled
and residue was recrystallised with isopropyl ether. 1.1 g of 17-α-hydroxyl-19-
norprogesteron-17-capronate was obtained, melting point 123°-124°C.
2. 2.0 g 3-methoxy-17α-hydroxy-17β-acetyl-δ2,5(10)-oestradien, 60 ml
capronic acid anhydride, 2.6 g p-toluensulfonate and 18.0 g water were mixed
and left for 6 h at room temperature. Then solution obtained was treated
ether and sodium bicarbonate and washed with water. Etheral solution was
dried over sodium sulfate. After distillation of ether 3.1 g 3,17α-dihydroxy-
δ3,5-19-norpregnadien-3,17-dicapronate was produced.
To solution of 3.1 g 3,17α-dihydroxy-δ3,5-19-norpregnadien-3,17-dicapronate
in 250 ml methanol 2.5 g conc. hydrochloric acid were added and mixture was
left for 1 h. Then mixture was filtered and residue was washed. After
recrystallisation with isopropyl ether 17-α-hydroxyl-19-norprogesteron-17-
capronate was obtained, melting point 121°-123°C.
Therapeutic Function
Progestin
Metabolism
Not Available
Properties of GESTONORONE CAPROATE
| Melting point: | 123-124° |
| Boiling point: | 453.43°C (rough estimate) |
| alpha | D +13° (chloroform) |
| Density | 1.11 |
| refractive index | 1.4840 (estimate) |
| storage temp. | Refrigerator |
| solubility | Chloroform (Slightly), Ethyl Acetate (Slightly), Methanol (Slightly) |
| form | Solid |
| color | Off-White to Pale Beige |
Safety information for GESTONORONE CAPROATE
Computed Descriptors for GESTONORONE CAPROATE
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloropyridazine 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 6-Methyl-2-pyridinemethanol 2-Pyridinecarboxaldehdye tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 1-Indanone 2-Chloromethyl-6-methyl-pyridine 4-Morpholinoaniline Abiraterone Ethyl Ether 1-nitro-3,5-dimethyl adamantine (NMEM) Vardenafil Bis-sulphonamide(Dimer) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran

You may like
-
![3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
2304754-51-4 -
 1126-74-5 99%+View Details
1126-74-5 99%+View Details
1126-74-5 -
 2-Fluoro-6-iodobenzoic acid 99%+View Details
2-Fluoro-6-iodobenzoic acid 99%+View Details
111771-08-5 -
 120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 -
 2-pyridineacetonitrile 2739-97-1 99%+View Details
2-pyridineacetonitrile 2739-97-1 99%+View Details
2739-97-1 -
 4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
138163-08-3 -
![ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
313544-31-9 -
 1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
91419-52-2
