dimazole
- CAS NO.:95-27-2
- Empirical Formula: C15H23N3OS
- Molecular Weight: 293.43
- MDL number: MFCD00602294
- EINECS: 202-406-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-20 17:00:22
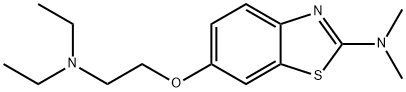
What is dimazole?
Originator
Aterola,Roche,US,1951
Background
Dimazole (diamthazole) is an antifungal. It was withdrawn in Franch in 1972 due to neuropsychiatric reactions.
Definition
ChEBI: 6-[2-(diethylamino)ethoxy]-N,N-dimethyl-1,3-benzothiazol-2-amine is a member of benzothiazoles.
Manufacturing Process
19.4 g of 2-dimethylamino-6-hydroxybenzothiazole (MP 245°C) were sludged
in a 500 cc three-necked flask with 250 cc of chlorbenzene. Then 4.4 g of
sodium hydroxide flakes were added and the mixture heated with agitation to
90°C. 4 cc of water were dropped in, and the mixture then heated slowly to
the boil while about 500 cc of the water-containing chlorbenzene were distilled
off. 50 cc of dry chlorbenzene were then added and the distillation was
continued until about 30 cc of the chlorbenzene were distilled off. The residue
was the sodium salt of thiazole in chlorbenzene. To the residue were added at
90°C, 15 g of fresh distilled 1-diethylamino-2-chloroethane. The mixture was
then refluxed at 133°C for three hours, then cooled to 35°C. 75 cc of water
and 5 cc of (40% by volume) sodium hydroxide solution were added and the
mixture stirred for one hour. The chlorbenzene layer which contained the
reaction product was separated from the aqueous layer in a separatory funnel.
The chlorbenzene solution was then dried with sodium sulfate for twelve
hours. It was then filtered and HCl gas was passed into the chlorbenzene
solution until saturated, while cooling and stirring. The dihydrochloride
precipitated as a white crystalline, sandy powder. The precipitate was filtered
and washed on the funnel with benzene and finally washed with ether. The
filter cake was dried at 80°C to 90°C. The 2-dimethylamino-6-(βdiethylaminoethoxy)-benzothiazole dihydrochloride thus obtained is a white
crystalline powder, MP 240°C to 243°C. It can be recrystallized from ethanol
and ether, or methanol or acetone.
The free base, which is an oil, can be obtained from the aqueous solution of
the dihydrochloride by adding dilute sodium hydroxide or sodium carbonate
solution. The base is soluble in ether, methanol, ethanol, benzene and the
like, but slightly soluble in water.
brand name
Asterol [as dihydrochloride] [Veterinary] (Hoffmann- LaRoche);Atelora;Aterola;Kesten;Mycotol.
Therapeutic Function
Antifungal
World Health Organization (WHO)
Dimazole, an antifungal agent, was introduced in 1951 for the treatment of tinea infections. Although the major manufacturer subsequently discontinued marketing preparations in the United States, the US Food and Drug Administration formally withdrew marketing approval for such preparations in 1977 on the grounds of their association with severe neurotoxic reactions, their potential for misuse and the availability of safer alternative products. Topical preparations of dimazole remain available in some 40 countries.
Metabolism
Not Available
Properties of dimazole
| Boiling point: | 195-200 °C(Press: 1 Torr) |
| Density | 1.137±0.06 g/cm3(Predicted) |
| pka | 9.49±0.25(Predicted) |
Safety information for dimazole
Computed Descriptors for dimazole
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran

You may like
-
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
