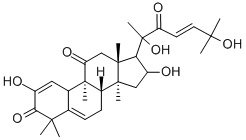
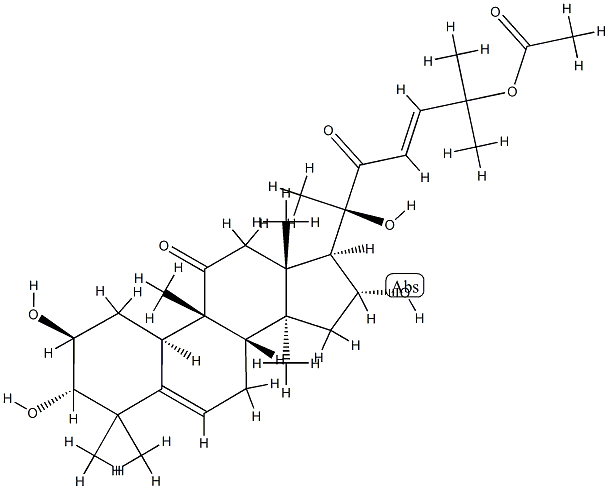
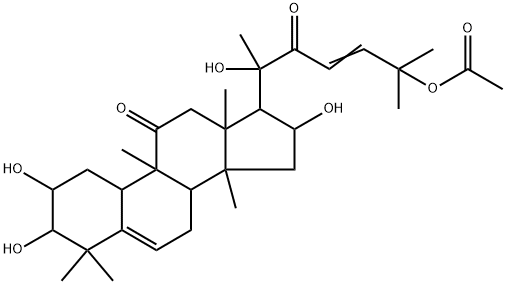
Cucurbitacin E
Synonym(s):α-Elaterin;α-Elaterine
- CAS NO.:18444-66-1
- Empirical Formula: C32H44O8
- Molecular Weight: 556.69
- MDL number: MFCD00135936
- EINECS: 242-325-2
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-11 15:21:01
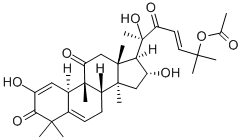
What is Cucurbitacin E?
Description
Cucurbitacin E is a plant-derived triterpene that has diverse biological activities. At a concentration of 10 pM, it reduces MPP+-induced death of neuronal PC12 cells through inhibition of autophagy in vitro. Cucurbitacin E inhibits growth of T24 bladder, MDA-MB-468 and MCF-7 breast, PC3 prostate, and colorectal cancer cell lines (IC50s = 50-1,000 nM) through induction of G2/M arrest and apoptosis. It increases bilirubin binding to human serum albumin (HSA) in human plasma in a dose-dependent manner. Cucurbitacin E also inhibits depolymerization of actin filaments isolated from rabbit skeletal muscle actin and in HeLa cells.
Chemical properties
white to beige powder
The Uses of Cucurbitacin E
Cucurbitacin E is a biochemical compound from the family of Cucurbitacins. Cucurbitacin E is a highly oxidated steroid consisting of a tetracyclic triterpene. Cucurbitacin E is known to possess broad spectrum of potential anti-inflammatory, antitumor andantioxidant effects.
The Uses of Cucurbitacin E
Cucurbitacin E has been used as a cofilin inhibitor. It is also used as a F-actin stabilizer to prevent membrane-associated periodic skeleton (MPS) loss and protect from axonal fragmentation.
Definition
ChEBI: A cucurbitacin in which a lanostane skeleton is multi-substituted with hydroxy, methyl and oxo substituents, with unsaturation at positions 1, 5 and 23.
General Description
This substance is a primary reference substance with assigned absolute purity (considering chromatographic purity, water, residual solvents, inorganic impurities). The exact value can be found on the certificate. Produced by PhytoLab GmbH & Co. KG.
Biochem/physiol Actions
Cucurbitacin E is a potent inhibitor of actin depolymerization. Cucurbitacin E is more active than jasplakinolide, and has a different mechanism of action, binding to a different site. Cucurbitacin E binds specifically to filamentous actin (F-actin) forming a covalent bond at residue Cys257, but not to monomeric actin (G-actin), stabilizing F-actin, without affecting actin polymerization or nucleation.
Properties of Cucurbitacin E
| Melting point: | 228-234°C |
| Boiling point: | 545.56°C (rough estimate) |
| alpha | D -59° (c = 0.7 in chloroform) |
| Density | 1.1059 (rough estimate) |
| refractive index | 1.4900 (estimate) |
| storage temp. | -20°C |
| solubility | DMSO: soluble15mg/mL, clear |
| form | powder |
| pka | 8.51±0.70(Predicted) |
| color | white to beige |
| optical activity | [α]/D -60 to -75°, c = 0.7 (CDCl3) |
Safety information for Cucurbitacin E
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral |
Computed Descriptors for Cucurbitacin E
| InChIKey | NDYMQXYDSVBNLL-MUYMLXPFSA-N |
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran


You may like
-
 Cucurbitacin E CAS 18444-66-1View Details
Cucurbitacin E CAS 18444-66-1View Details
18444-66-1 -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5
