Cefmetazole sodium
- CAS NO.:56796-39-5
- Empirical Formula: C15H18N7NaO5S3
- Molecular Weight: 495.52
- MDL number: MFCD00214260
- EINECS: 627-393-7
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-28 00:01:47
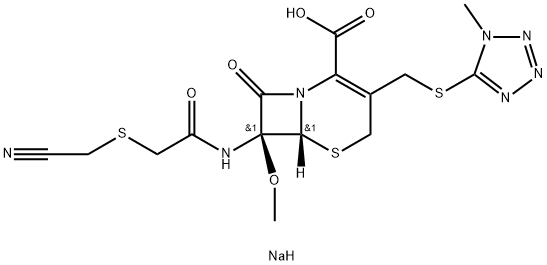
What is Cefmetazole sodium?
Chemical properties
White Solid
The Uses of Cefmetazole sodium
Cefmetazole sodium is used clinically in combination with Tanreqing for the treatment of Pulmonary infections like Senile Emphysema and other acute bacterial infections.
The Uses of Cefmetazole sodium
Semi-synthetic antibiotic derived from Cephamycin. Antibacterial
What are the applications of Application
Cefmetazole sodium salt is a cephalosporin antibiotic
Definition
ChEBI: An organic sodium salt that is the sodium salt of cefmetazole.
brand name
Zefazone (Pharmacia & Upjohn).
Clinical Use
Cefmetazole (Zefazone) is a semisynthetic, third-generation,parenteral cephalosporin of the cephamycin group. Likeother cephamycins, the presence of the 7α-methoxyl groupconfers resistance to many β-lactamases. Cefmetazoleexhibits significantly higher potency against members ofthe Enterobacteriaceae family but lower activity againstBacteroides spp. than cefoxitin. It is highly active againstN. gonorrhoeae, including -lactamase–producing strains. Incommon with other cephamycins, cefmetazole is ineffectiveagainst indole-positive Proteus, Enterobacter, Providencia,Serratia, and Pseudomonas spp. Cefmetazole has the MTTmoiety associated with increased bleeding in certain highriskpatients. It has a plasma half-life of 1.1 hours.
Properties of Cefmetazole sodium
| Melting point: | >130°C (dec.) |
| storage temp. | Inert atmosphere,2-8°C |
| solubility | H2O: soluble50mg/mL |
| form | powder |
| color | White to Off-White |
| Water Solubility | Soluble in water |
| Sensitive | Hygroscopic |
| Merck | 14,1926 |
| Stability: | Hygroscopic |
| CAS DataBase Reference | 56796-39-5(CAS DataBase Reference) |
Safety information for Cefmetazole sodium
| Signal word | Danger |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07  Health Hazard GHS08 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H317:Sensitisation, Skin H319:Serious eye damage/eye irritation H334:Sensitisation, respiratory H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Cefmetazole sodium
| InChIKey | BITQGIOJQWZUPL-PBCQUBLHSA-M |
New Products
Pentadecanoic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 2-Bromo-5-Iodopyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile Tetrahydropyranyl-4-acetic acid Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside 5-fluoro-1,3-benzodioxole 2-Bromo-6-fluoroaniline 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-oneRelated products of tetrahydrofuran


You may like
-
 Cefmetazole sodium 95% CAS 56796-39-5View Details
Cefmetazole sodium 95% CAS 56796-39-5View Details
56796-39-5 -
 Cefmetazole Sodium Salt CAS 56796-39-5View Details
Cefmetazole Sodium Salt CAS 56796-39-5View Details
56796-39-5 -
 Cefmetazole sodium salt CAS 56796-39-5View Details
Cefmetazole sodium salt CAS 56796-39-5View Details
56796-39-5 -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
