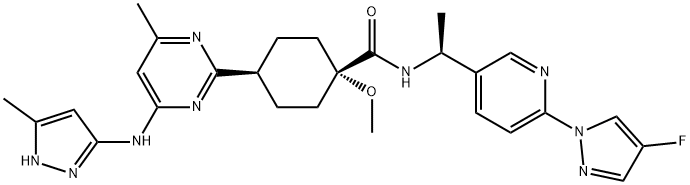
Pralsetinib
- CAS NO.:2097132-94-8
- Empirical Formula: C27H32FN9O2
- Molecular Weight: 533.6
- MDL number: MFCD31810155
- Update Date: 2026-04-22 18:57:36
What is Pralsetinib?
Absorption
Pralsetinib given at 400 mg once daily resulted in a mean steady-state Cmax of 2830 ng/mL (coefficient of variation, CV, 52.5%) and AUC0-24h of 43900 ng*h/mL (CV 60.2%). The Cmax and AUC of pralsetinib increased inconsistently with increasing doses between 60 and 600 mg once daily, with a median Tmax across this range of between two and four hours. At 400 mg once daily, pralsetinib reached steady-state plasma concentration by three to five days.
Pralsetinib absorption is affected by food. A single dose of 400 mg given with a high-fat meal (800 to 1000 calories with 50 to 60% of calories coming from fat) increased the mean Cmax by 104% (95% CI 65-153%), mean AUC0-∞ by 122% (95% CI 96-152%), and the median Tmax from four to 8.5 hours.
Toxicity
Pralsetinib administered to rats at 20 mg/kg (roughly 2.5-3.6 times the recommended human exposure) resulted in resorption of litters in pregnant female mice in 92% of pregnancies (82% complete resorption); resorption occurred at doses as low as 5 mg/kg (0.3 times the recommended human exposure). Both male and female rats given 10 mg/kg pralsetinib or more had observable degeneration within the testis/ovaries. In 28-day rat and monkey studies, once-daily pralsetinib resulted in histological necrosis at doses 1.1 or more times the recommended human dose and myocardial hemorrhage at doses 2.6 or more times the recommended human dose. Also, pralsetinib induced hyperphosphatemia (rats only, dose 2.4-3.5 times the recommended human dose) and multi-organ mineralization (dose 0.11 or more times the recommended human dose).
Description
Pralsetinib is a potent, selective RET inhibitor, and was optimized from a hit compound identified from a compound library that included more than 60 chemical scaffolds. However, as of 2020, detailed medicinal chemistry on the optimization process has yet to be published. Pralsetinib proved to be more potent and selective than cabozantinib or vandertanib against both wild-type and abnormal RET. The agent also showed a level of selectivity against RET relative to VEGFR2, whereas previous agents showed very little selectivity. On September 4, 2020, the Food and Drug Administration granted accelerated approval to pralsetinib (GAVRETO®, Blueprint Medicines Corporation) for adult patients with metastatic RET fusion-positive non-small cell lung cancer (NSCLC) as detected by an FDA-approved test.
The Uses of Pralsetinib
Pralsetinib is a highly potent and selective RET inhibitor designed for RET-driven cancers.
Background
Pralsetinib, similar to the previously approved selpercatinib, is a kinase inhibitor with enhanced specificity for RET tyrosine kinase receptors (RTKs) over other RTK classes. Enhanced RET (Rearranged during transfection) oncogene expression is a hallmark of many cancers, including non-small cell lung cancer. Although multikinase inhibitors, including cabozantinib, ponatinib, sorafenib, sunitinib, and vandetanib, have shown efficacy in RET-driven cancers, their lack of specificity is generally associated with substantial toxicity. Pralsetinib (BLU-667) and selpercatinib (LOXO-292) represent the first generation of specific RET RTK inhibitors for the treatment of RET-driven cancers.
Although a phase 1/2 trial of pralsetinib termed ARROW (NCT03037385) is still ongoing, pralsetinib was granted accelerated FDA approval on September 4, 2020, for the treatment of metastatic RET-fusion positive non-small cell lung cancer. It is currently marketed under the brand name GAVRETO? by Blueprint Medicines.
Indications
Pralsetinib is indicated for the treatment of metastatic non-small cell lung cancer (NSCLC) in adult patients who are confirmed to possess a rearranged during transfection (RET) gene fusion, as determined by an FDA approved test. It is also indicated in adult and pediatric patients 12 years of age and older for the treatment of advanced or metastatic RET fusion-positive thyroid cancer who require systemic therapy and for whom radioactive iodine is not appropriate. The indication for advanced or metastatic RET fusion-positive thyroid cancer was approved under accelerated approval based on the overall response rate and duration of response, and continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
Indications
Pralsetinib is the second selective RET inhibitor approved by the FDA (following Lilly's selpercatinib) for the treatment of adult patients with metastatic RET (rearranged during transfection) fusion-positive non-small cell lung cancer (NSCLC); (2) adult and pediatric patients >=12 years of age with advanced or metastatic RET-mutant medullary thyroid cancer (MTC) who require systemic therapy; and (3) adult and pediatric patients >=12 years of age with advanced or metastatic RET fusion-positive thyroid cancer who require systemic therapy and who are radioactive iodine-refractory.
brand name
Gavreto
Synthesis Reference(s)
[1] GAIKWAD RAJENDRA. Green One-Pot Chemo-Enzymatic Synthesis of a Key Chiral Amine Intermediate: Useful to Pralsetinib Synthesis[J]. ChemistrySelect, 2023. DOI:10.1002/slct.202204409.
[2] HUGHES* D L. Review of Synthetic Routes and Crystalline Forms of the Oncology Drugs Capmatinib, Selpercatinib, and Pralsetinib[J]. Organic Process Research & Development, 2021. DOI:10.1021/acs.oprd.1c00282.
General Description
Class: receptor tyrosine kinase; Treatment: RET-altered lung, thyroid cancers; Other name: BLU-667; Elimination half-life = 22 h; Protein binding = 97%
Pharmacokinetics
Pralsetinib exerts an anti-tumour effect through specific inhibition of the rearranged during transfection (RET) tyrosine kinase, including multiple distinct oncogenic RET fusions, mutated RET kinase domains harbouring gatekeeper mutations, and in RET kinases with a variety of activating single point mutations. Due to pralsetinib's high selectivity for RET over other kinases, both in vitro and in vivo, pralsetinib has been described as having a better safety profile compared to previously used multi-kinase inhibitors. Despite this, pralsetinib use may increase the risk of hypertension, hemorrhagic events, impaired wound healing, hepatotoxicity, interstitial lung disease/pneumonitis, and embryo-fetal toxicity.
Side Effects
Pralsetinib may cause side effects: Nausea, vomiting, loss of appetite, diarrhea, constipation, extreme tiredness, dizziness, weakness, night sweats, rapid heartbeat, heartburn, shortness of breath, headache, sores in the mouth, confusion, changes in vision, fever, cough, chills, nosebleeds, pale skin, rash, itching, hives, weight changes, hair loss, back pain, muscle pain, joint pain, bone pain, unusual bruising or bleeding, blood in the urine or stool, bloody or black tarry stools, difficulty falling asleep or staying asleep, unusual vaginal bleeding, and other less common effects.
Metabolism
Pralsetinib is metabolized in vitro primarily by CYP3A4 and to a lesser extent by CYP2D6 and CYP1A2. Pralsetinib given as a single oral dose of 310 mg in healthy volunteers led to the detection of metabolites from both oxidation (M453, M531, and M549b) and glucuronidation (M709), although these constituted less than 5% of the detected material.
Mode of action
Kinase inhibitor of wild-type RET, oncogenic RET fusions, and select mutations. Pralsetinib may also inhibit other pathways including those through FLT3, JAK1-2, PDGFRB, VEGFR-2, and FGFR1. RET fusion proteins and activating point mutations can act as oncogenic drivers by promoting cell proliferation of tumor cell lines and pralsetinib inhibits this process.
Properties of Pralsetinib
| Boiling point: | 799.1±60.0 °C(Predicted) |
| Density | 1.40±0.1 g/cm3(Predicted) |
| storage temp. | Store at 4°C |
| solubility | DMSO : ≥ 100 mg/mL (187.41 mM);Water : < 0.1 mg/mL (insoluble) |
| form | Solid |
| pka | 14.33±0.10(Predicted) |
| color | White to off-white |
Safety information for Pralsetinib
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Pralsetinib
| InChIKey | GBLBJPZSROAGMF-BATDWUPUSA-N |
| SMILES | [C@]1(OC)(C(N[C@H](C2=CC=C(N3C=C(F)C=N3)N=C2)C)=O)CC[C@H](C2=NC(NC3C=C(C)NN=3)=CC(C)=N2)CC1 |
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloropyridazine 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 6-Methyl-2-pyridinemethanol 2-Pyridinecarboxaldehdye tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 1-Indanone 2-Chloromethyl-6-methyl-pyridine 4-Morpholinoaniline Abiraterone Ethyl Ether 1-nitro-3,5-dimethyl adamantine (NMEM) Vardenafil Bis-sulphonamide(Dimer) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran
You may like
-
![3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
2304754-51-4 -
 1126-74-5 99%+View Details
1126-74-5 99%+View Details
1126-74-5 -
 2-Fluoro-6-iodobenzoic acid 99%+View Details
2-Fluoro-6-iodobenzoic acid 99%+View Details
111771-08-5 -
 120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 -
 2-pyridineacetonitrile 2739-97-1 99%+View Details
2-pyridineacetonitrile 2739-97-1 99%+View Details
2739-97-1 -
 4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
138163-08-3 -
![ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
313544-31-9 -
 1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
91419-52-2