Avapritinib
- CAS NO.:1703793-34-3
- Empirical Formula: C26H27FN10
- Molecular Weight: 498.56
- MDL number: MFCD31544325
- Update Date: 2026-04-24 14:58:33
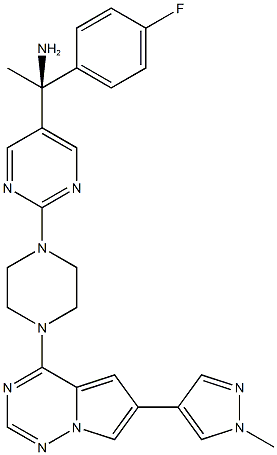
What is Avapritinib?
Absorption
A 300mg oral dose of avapritinib reaches a Cmax of 813ng/mL with a Tmax of 2.0-4.1h and an AUC of 15400h*ng/mL.
Toxicity
Data regarding overdoses of avapritinib are not readily available.
Description
BLU-285 is a dual inhibitor of KIT receptor and platelet-derived growth factor receptor α (PDGFRα) tyrosine kinases with activating loop mutations (IC50s = 0.27 and 0.24 nM for KITD816V and PDGFRαD842V, respectively). It is >150-fold selective for KITD816V and PDGFRαD842V over a kinase panel at a concentration of 3 μM. BLU-285 also has activity against a panel of KIT and PDGFRα loop mutants identified in patients with gastrointestinal stromal tumors (GISTs; IC50s = <100 nM). It inhibits growth of cancer cell lines containing wild-type and mutant KIT and PDGFRα, demonstrating increased potency in cell lines expressing KIT and PDGFRα with activating loop mutations. BLU-285 (0.3-30 mg/kg) reduces tumor volume in a P815 KITD814Y mastocytoma allograft mouse model and a GIST patient-derived mouse xenograft model in a dose-dependent manner. Formulations containing BLU-285 have been used to treat KIT- and PDGFRα-driven malignancies.
The Uses of Avapritinib
Avapritinib is a medication used for the treatment of tumors due to one specific rare mutation. Avapritinib is a potent, selective, and orally active KIT and PDGFRA activation loop mutant kinases inhibitor.
Indications
Avapritinib is indicated for the treatment of adults with unresectable or metastatic GIST harboring a platelet-derived growth factor receptor alpha (PDGFRA) exon 18 mutation, including PDGFRA D842V mutations.
It is also used to treat adult patients with advanced systemic mastocytosis (AdvSM). AdvSM includes patients with aggressive systemic mastocytosis (ASM), systemic mastocytosis with an associated hematological neoplasm (SM-AHN), and mast cell leukemia. However, it is not recommended for the treatment of patients with AdvSM with platelet counts of less than 50 X 109 L.
Background
Avapritinib, or BLU-285, is a selective tyrosine kinase inhibitor of KIT and platelet derived growth factor receptor alpha indicated for the treatment of unresectable, metastatic gastrointestinal stromal tumors and advanced systemic mastocytosis. It is one of the first medications available for the treatment of multidrug resistant cancers. Avapritinib shares a similar mechanism with ripretinib.
Avapritinib was granted FDA approval on 9 January 2020 and EMA approval on 24 September 2020.
Indications
Avapritinib is approved for the treatment of adult patients with unresectable or metastatic gastrointestinal mesenchymal stromal tumour (GIST) carrying platelet-derived growth factor receptor A (PDGFRA) exon 18 mutations, including the D842V mutation, as well as adult patients with advanced systemic mastocytosis (ASM).
brand name
Ayvakit
General Description
Class: receptor tyrosine kinase; Treatment: GIST, SM; Other name: BLU-285; Elimination half-life = 32–57 h; Protein binding = 98.8%
Pharmacokinetics
Avapritinib is a selective kinase inhibitor that negatively modulates the action of cell transporters to resensitize them to other chemotherapies. It has a long duration of action as it is given once daily. Patients should be counselled regarding the risk of intracranial hemorrhage, CNS effects, and embryo-fetal toxicity.
Metabolism
Avapritinib is metabolized mainly by CYP3A4 and CYP2C9 in vitro. A 310mg oral dose is recovered as 49% unchanged drug, 35% hydroxy glucuronide metabolite, and 14% oxidatively deaminated metabolite.
Properties of Avapritinib
| Density | 1.42±0.1 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | DMSO:83.11(Max Conc. mg/mL);166.7(Max Conc. mM) |
| form | A crystalline solid |
| pka | 6.96±0.10(Predicted) |
| color | White to light yellow |
| CAS DataBase Reference | 1703793-34-3 |
Safety information for Avapritinib
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07  Health Hazard GHS08 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H317:Sensitisation, Skin H319:Serious eye damage/eye irritation H332:Acute toxicity,inhalation H334:Sensitisation, respiratory H340:Germ cell mutagenicity H350:Carcinogenicity H360:Reproductive toxicity H373:Specific target organ toxicity, repeated exposure H412:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P201:Obtain special instructions before use. P202:Do not handle until all safety precautions have been read and understood. P271:Use only outdoors or in a well-ventilated area. P272:Contaminated work clothing should not be allowed out of the workplace. P280:Wear protective gloves/protective clothing/eye protection/face protection. P285:In case of inadequate ventilation wear respiratory protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P304+P340:IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P308+P313:IF exposed or concerned: Get medical advice/attention. P342+P311:IF experiencing respiratory symptoms: call a POISON CENTER or doctor/physician. P403:Store in a well-ventilated place. |
Computed Descriptors for Avapritinib
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloropyridazine 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 6-Methyl-2-pyridinemethanol 2-Pyridinecarboxaldehdye tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 1-Indanone 2-Chloromethyl-6-methyl-pyridine 4-Morpholinoaniline Abiraterone Ethyl Ether 1-nitro-3,5-dimethyl adamantine (NMEM) Vardenafil Bis-sulphonamide(Dimer) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran
You may like
-
![3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
2304754-51-4 -
 1126-74-5 99%+View Details
1126-74-5 99%+View Details
1126-74-5 -
 2-Fluoro-6-iodobenzoic acid 99%+View Details
2-Fluoro-6-iodobenzoic acid 99%+View Details
111771-08-5 -
 120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 -
 2-pyridineacetonitrile 2739-97-1 99%+View Details
2-pyridineacetonitrile 2739-97-1 99%+View Details
2739-97-1 -
 4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
138163-08-3 -
![ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
313544-31-9 -
 1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
91419-52-2