N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine
- CAS NO.:477600-73-0
- Empirical Formula: C20H25N5
- Molecular Weight: 335.45
- MDL number: MFCD16877475
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-07-04 14:23:51
![N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine Structural](https://img.chemicalbook.in/CAS/GIF/477600-73-0.gif)
What is N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine?
The Uses of N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine
N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine is an important pharmaceutical intermediate used for the synthesis of tofacitinib. Tofacitinib is a Janus kinase (JAK) inhibitor drug used to treat rheumatoid arthritis and psoriatic arthritis.
Synthesis
N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine is prepared by the reaction of 4-chloropyrrolopyrimidine and (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3-amine dihydrochloride. The specific synthesis steps are as follows:
The reaction flask is added to sodium hydroxide (10.0g, 250mmol), water and dissolved, make 10% of the solution, adding acetone to another reaction bottle 80g, the toluene sulfonyl chloride (38.13g, 200mmol), after stirring to dissolve, then adding 4-chloro pyrrolo pyrimidine (15.36g, 100mmol), stirring mixing, cooling to 0 °C the following, dropping sodium hydroxide solution, the temperature is controlled at 5 °C the following, completion of the dropping, heating, to control the temperature to 20-30 °C stirring within the range of the reaction, to the reaction end after TLC monitoring, filtering, to get the product into the reaction bottle, adding water 200 ml, (3R, 4R)-cis-1-benzyl-4-methyl-3-methylamino-piperidine dihydrochloride (21.33g, 105mmol), stirring to dissolve, then adding potassium carbonate (82.93g, 600mmol), stirring, heating 95 °C, TLC monitoring to the reaction end of the, cooling to 45-55°C, by adding acetonitrile, preserving heat and stirring 1 hour, cooling to room temperature, crystallization, filtration, washing, the wet articles in added in the reaction bottle, adding dimethyl sulfoxide 250 ml, by adding 50% sodium hydroxide solution to 250 ml, stirring and heating 95 °C, TLC monitoring to the reaction end rear, layered, water extraction once with dimethyl sulfoxide, merger dimethyl asian sulphone level, cooling to 75-85°C, water slowly under stirring, the stirring cooling to room temperature, filtering, washing, 50% ethanol washing, filtering, drying, be [(3R, 4R) - 1 benzyl-4-methyl-piperidin-3-yl]-methyl-(7H-pyrrolo [2,3-d] pyrimidin-4-yl)-amine 25.83g, yield 77.0%, optical purity 99.8% (HPLC method).![N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine synthesis N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine synthesis](/NewsImg/2024-01-11/6384057056171848491778669.png)
Properties of N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine
| Melting point: | 67-69℃ |
| Density | 1.205±0.06 g/cm3(Predicted) |
| storage temp. | under inert gas (nitrogen or Argon) at 2–8 °C |
| pka | 13.36±0.50(Predicted) |
Safety information for N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine
Computed Descriptors for N-((3R,4R)-1-benzyl-4-methylpiperidin-3-yl)-N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine
New Products
DL-beta-(3-Bromophenyl)alanine Tetrabutylammonium perchlorate N,O-Dimethylhydroxylamine hydrochloride (RS)-beta-Amino-beta-(4-bromophenyl)propionic acid 2-Amino-5-bromo-4-(trifluoromethyl)pyridine(RM for Indian lab) N,N CARBONYL DIIMIDAZOLE Levothyroxine Impurity-F Montelukast EP Impurity-D/Montelukast USP Related Compound C Atorvastatin FXA Impurity/Atorvastatin Cyclic 6-Hydroxy Impurity Sodium Salt Isosulfan blue Keto N-Oxide Impurity Ivermectin Impurity F N-Nitroso des Methyl Tramadol/N-Nitroso-N-Desmethyl-Tramadol (R)-BoroLeu-(+)-Pinanediol-CF3COOH 1,4-bis(methylsulfonyl)butane 4-(5-amino-1-methyl-1h-benzoimidazol-2-yl)-butyric acid isopropyl ester. 5-Methyl-1,3-benzenediacetonitrile 4-Fluorothiophenol 1-methyl amino-2,4-dinitro benzene CSA (DL-10-Camphorsulfonic Acid) 2-Hydroxy-4-methylnicotinic acid 3-iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine (2S)-1-((2S,3S)-3-(2-methylbutyl)-4-oxooxetan-2-yl)pentadecan-2-yl formylleucinate 1-(3,5-dichlorophenyl)-2,2,2-trifluoroethane-1-sulfonyl chloride S-(1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl) ethanethioateRelated products of tetrahydrofuran

![N-methyl-N-((3R,4R)-4-methylpiperidin-3-yl)-7H-pyrrolo[2,3-d]pyrimidin-4-amine](https://img.chemicalbook.in/CAS/GIF/477600-74-1.gif)
![4-CHLORO-7-TOSYL-7H-PYRROLO[2,3-D]PYRIMIDINE](https://img.chemicalbook.in/CAS/GIF/479633-63-1.gif)
![7H-Pyrrolo[2,3-d]pyriMidin-4-aMine, N-Methyl-N-[(3R,4R)-4-Methyl-1-(phenylMethyl)-3-piperidinyl]-7-[(4-Met hylphenyl)sulfonyl]-](https://img.chemicalbook.in/CAS/20150408/GIF/923036-30-0.gif)
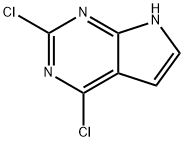
![4-Chloro-7H-pyrrolo[2,3-d]pyrimidine](https://img.chemicalbook.in/CAS/GIF/3680-69-1.gif)
![3-PiperidinaMine, 1-acetyl-N,4-diMethyl-N-1H-pyrrolo[2,3-d]pyriMidin-4-yl-, (3R,4R)- (9CI)](https://img.chemicalbook.in/CAS/20150408/GIF/477600-76-3.gif)
![3-((3S,4R)-4-Methyl-3-(Methyl(7H-pyrrolo[2,3-d]pyriMidin-4-yl)aMino)piperidin-1-yl)-3-oxopropanenitrile](https://img.chemicalbook.in/CAS/GIF/1092578-48-7.gif)
You may like
-
![[(3R,4R)-1-benzyl-4-methyl-piperidin-3-yl]-methyl-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-amine 98%](https://img.chemicalbook.in//ProductImageIndia/2024-03/Raw/6e57a743-4832-4f97-95bf-b142073c11b6.png) [(3R,4R)-1-benzyl-4-methyl-piperidin-3-yl]-methyl-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-amine 98%View Details
[(3R,4R)-1-benzyl-4-methyl-piperidin-3-yl]-methyl-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-amine 98%View Details -
![N-((3R,4R)-1-Benzyl-4-methylpiperidin-3-yl)-n-methyl-7h-pyrrolo[2,3-d]pyrimidin-4-amine 96% CAS 477600-73-0](https://img.chemicalbook.in//Content/image/CP5.jpg) N-((3R,4R)-1-Benzyl-4-methylpiperidin-3-yl)-n-methyl-7h-pyrrolo[2,3-d]pyrimidin-4-amine 96% CAS 477600-73-0View Details
N-((3R,4R)-1-Benzyl-4-methylpiperidin-3-yl)-n-methyl-7h-pyrrolo[2,3-d]pyrimidin-4-amine 96% CAS 477600-73-0View Details
477600-73-0 -
 674783-97-2 98+View Details
674783-97-2 98+View Details
674783-97-2 -
 H-D-TRP(FOR)-OH HCL 98+View Details
H-D-TRP(FOR)-OH HCL 98+View Details
367453-01-8 -
 49830-37-7 98+View Details
49830-37-7 98+View Details
49830-37-7 -
 3-Amino-3-(4-fluorophenyl)propanoic acid 98+View Details
3-Amino-3-(4-fluorophenyl)propanoic acid 98+View Details
325-89-3 -
 1428243-26-8 98+View Details
1428243-26-8 98+View Details
1428243-26-8 -
 1-aminocyclopentane carbonitrile, HCl 98+View Details
1-aminocyclopentane carbonitrile, HCl 98+View Details
16195-83-8
