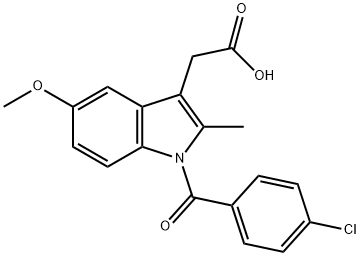
5-METHOXY-2-METHYLINDOLE
Synonym(s):NSC 63817
- CAS NO.:1076-74-0
- Empirical Formula: C10H11NO
- Molecular Weight: 161.2
- MDL number: MFCD00005620
- EINECS: 214-066-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 14:59:00
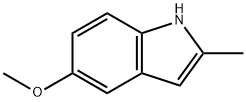
What is 5-METHOXY-2-METHYLINDOLE?
Description
5-methoxy-2-methylindole is a useful organic raw material. It can be used as the reactant in the preparation of indolylquinoxalines by condensation reactions, in preparation of alkylindoles via Ir-catalyzed reductive alkylation, in arylation reactions in the presence of a palladium acetate catalyst, in enantioselective Friedel-Crafts alkylation, as well as in stereo selective synthesis of cyclopentaindolones via stereoselective [3+2] cyclopentannulation. It is also employed in inhibiting the chlorinating activity of myeloperoxidase (MPO) and in the metabolic synthesis of acrylacetic acid anti-inflammatory synthesis.
Chemical properties
white to light beige crystalline powder or
The Uses of 5-METHOXY-2-METHYLINDOLE
An indole derivative shown to be an effective inhibitor of the chlorinating activity of myeloperoxidase (MPO). Used in the metabolic synthesis of arylacetic acid antiinflammatory synthesis.
The Uses of 5-METHOXY-2-METHYLINDOLE
reactant in preparation of indolylquinoxalines by condensation reactions1reactant in preparation of alkylindoles via Ir-catalyzed reductive alkylation2reactant in arylation reactions using a palladium acetate catalyst3reactant in enantioselective Friedel-Crafts alkylation4reactant in stereoselective synthesis of cyclopentaindolones via stereoselective [3+2] cyclopentannulation5
The Uses of 5-METHOXY-2-METHYLINDOLE
It is employed as a reactant in preparation of indolylquinoxalines by condensation reactions, reactant in preparation of alkylindoles via Ir-catalyzed reductive alkylation, reactant in arylation reactions using a palladium acetate catalyst, reactant in enantioselective Friedel-Crafts alkylation and reactant in stereoselective synthesis of cyclopentaindolones via stereoselective [3+2] cyclopentannulation. As an indole derivative shown to be an effective inhibitor of the chlorinating activity of myeloperoxidase (MPO). Used in the metabolic synthesis of arylacetic acid anti-inflammatory synthesis.
Definition
ChEBI: 5-Methoxy-2-methyl-1H-indole is a member of indoles.
References
https://www.alfa.com/en/catalog/B21348/
http://www.sigmaaldrich.com/catalog/product/aldrich/m15451?lang=en®ion=US
https://pubchem.ncbi.nlm.nih.gov/compound/5-Methoxy-2-methylindole#section=2D-Structure
Properties of 5-METHOXY-2-METHYLINDOLE
| Melting point: | 86-88 °C (lit.) |
| Boiling point: | 145 °C / 1.5mmHg |
| Density | 1.0840 (rough estimate) |
| refractive index | 1.5200 (estimate) |
| storage temp. | Keep in dark place,Sealed in dry,Room Temperature |
| solubility | Soluble in methanol (very faint turbidity.) |
| form | Crystalline Powder or Crystals |
| pka | 17.28±0.30(Predicted) |
| color | White to light beige |
| CAS DataBase Reference | 1076-74-0(CAS DataBase Reference) |
Safety information for 5-METHOXY-2-METHYLINDOLE
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H332:Acute toxicity,inhalation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for 5-METHOXY-2-METHYLINDOLE
| InChIKey | VSWGLJOQFUMFOQ-UHFFFAOYSA-N |
New Products
5-Chlorothiophene-2-carboxylic acid 2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-3-methyl xanthine Maleic hydrazide 6-Methyl-2-(p-tolyl)imidazo[1,2-a]pyridine 3,4-Dihydroxyacetophenone 3,4-Dimethoxyacetophenone 4-Nitroacetophenone DL-Glutamic acid 3-Nitroacetophenone 4'-Chloro-3'-nitroacetophenone 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 1-Boc-4-cyanopiperidine 2-Fluoro-6-iodobenzoic acid 3-Pyridineacrylic acid 3-Hydroxy-2-methylbenzoic acid 1,3-Dibromo-5-fluorobenzene 2-((2,6-Difluorobenzyl)(ethoxycarbonyl)amino)-4-((dimethylamino)methyl)-5-(4-nitrophenyl)thiophene-3-carboxylic acid [N(E)]-N-Ethylidene-2-methyl-2-propanesulfinamide 4-Isopropyl Thiophenol 6-(chloromethyl)pteridine-2,4-diamine monohydrochloride 2-Chlorophenyl diphenyl chloromethaneRelated products of tetrahydrofuran
You may like
-
 5-Methoxy-2-methylindole CAS 1076-74-0View Details
5-Methoxy-2-methylindole CAS 1076-74-0View Details
1076-74-0 -
 5-Methoxy-2-methylindole CAS 1076-74-0View Details
5-Methoxy-2-methylindole CAS 1076-74-0View Details
1076-74-0 -
 586-37-8 98+View Details
586-37-8 98+View Details
586-37-8 -
 1-Boc-Azetidine-3-yl-methanol 98+View Details
1-Boc-Azetidine-3-yl-methanol 98+View Details
142253-56-3 -
 1197-09-7' 98+View Details
1197-09-7' 98+View Details
1197-09-7' -
 3,4-Dimethoxyacetophenone 98+View Details
3,4-Dimethoxyacetophenone 98+View Details
1131-62-0 -
 4-Acetylbiphenyl 92-91-1 98+View Details
4-Acetylbiphenyl 92-91-1 98+View Details
92-91-1 -
 99-93-4 98+View Details
99-93-4 98+View Details
99-93-4
