18-HYDROXY-11-DEOXYCORTICOSTERONE
- CAS NO.:379-68-0
- Empirical Formula: C21H30O4
- Molecular Weight: 346.46
- MDL number: MFCD00010486
- EINECS: 2068343
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-01-13 11:11:09
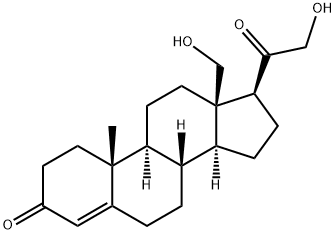
What is 18-HYDROXY-11-DEOXYCORTICOSTERONE?
Description
18-hydroxy-11-deoxy Corticosterone (18-OH-DOC) is a mineralocorticoid secreted by the zona fasciculata of the adrenal gland. Its biosynthesis is regulated by adrenocorticotropic hormone (ACTH; ) as well as angiotensin II , which increases 18-OH-DOC production in isolated human adrenal glomerulosa cells. 18-OH-DOC can be formed via conversion of 11-deoxy corticosterone (DOC; ) in human SK-MEL188 melanoma cells. 18-OH-DOC is an intermediate in the metabolism of progesterone and can be converted to aldosterone by the capsular portion of rat adrenal glands. Continuous infusion of 18-OH-DOC (200 μg/rat per day) increases systolic blood pressure in uninephrectomized saline-drinking rats. Plasma levels of 18-OH-DOC are elevated in a db/db mouse model of type 2 diabetes.
The Uses of 18-HYDROXY-11-DEOXYCORTICOSTERONE
11-Deoxy-18-hydroxycorticosterone is an analog of Corticosterone (C695700); a glucocorticoid and intermediate in the biosynthesis of Aldosterone (A514700) which is an adrenocortical steroid isolated from the adrenal cortex.
Definition
ChEBI: 18-hydroxydeoxycorticosterone is a 21-hydroxy steroid, a 3-oxo-Delta(4) steroid, a 20-oxo steroid and a 18-hydroxy steroid.
Purification Methods
Recrystallise 18-hydroxy-11-deoxycorticosterone from Et2O/Me2CO to give crystals m 200-205o. When it is recrystallised from M2CO, it has m 191-195o. It has UV with max at 240nm. The 21-O-acetoxy-18-hydroxy derivative has m 158-159o (from Et2O/*C6H6), and the 21-O-acetoxy-18,20-epoxy derivative has m 149-154o (from Et2O). [Kahnt et al. Helv Chim Acta 38 1237 1955; Pappo J Am Chem Soc 81 1010 1959.]
Properties of 18-HYDROXY-11-DEOXYCORTICOSTERONE
| Melting point: | 171-173 °C |
| storage temp. | 2-8°C |
| solubility | Chloroform (Slightly, Sonicated), Methanol (Slightly, Heated) |
| form | Solid |
| color | White to Off-White |
Safety information for 18-HYDROXY-11-DEOXYCORTICOSTERONE
Computed Descriptors for 18-HYDROXY-11-DEOXYCORTICOSTERONE
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloropyridazine 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 6-Methyl-2-pyridinemethanol 2-Pyridinecarboxaldehdye tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 1-Indanone 2-Chloromethyl-6-methyl-pyridine 4-Morpholinoaniline Abiraterone Ethyl Ether 1-nitro-3,5-dimethyl adamantine (NMEM) Vardenafil Bis-sulphonamide(Dimer) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran

![ALDOSTERONE, D [1,2,6,7-3H(N)]](https://img.chemicalbook.in/StructureFile/ChemBookStructure2/GIF/CB5458996.gif)


![ALDOSTERONE, D-[1,2-3H(N)]-](https://img.chemicalbook.in/StructureFile/ChemBookStructure2/GIF/CB5241895.gif)
You may like
-
![3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
2304754-51-4 -
 1126-74-5 99%+View Details
1126-74-5 99%+View Details
1126-74-5 -
 2-Fluoro-6-iodobenzoic acid 99%+View Details
2-Fluoro-6-iodobenzoic acid 99%+View Details
111771-08-5 -
 120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 -
 2-pyridineacetonitrile 2739-97-1 99%+View Details
2-pyridineacetonitrile 2739-97-1 99%+View Details
2739-97-1 -
 4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
138163-08-3 -
![ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
313544-31-9 -
 1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
91419-52-2
