Zabofloxacin
- CAS NO.:219680-11-2
- Empirical Formula: C19H20FN5O4
- Molecular Weight: 401.39
- MDL number: MFCD18633297
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-21 10:31:43
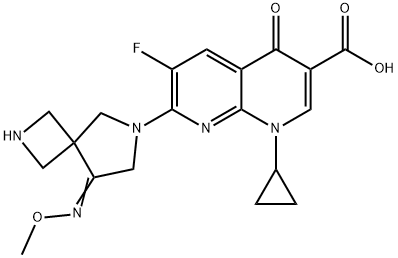
What is Zabofloxacin?
Description
Zabofloxacin is a quinolone antibiotic originally developed by Dong Wha Pharmaceuticals and licensed to Pacific Beach Biosciences in 2007. In March 2015, Korea’s Ministry of Food and Drug Safety (MFDS) approved zabofloxacin for the treatment of acute bacterial exacerbation of chronic obstructive pulmonary disease (ABE-COPD). In 2016, zabofloxacin gained approval from the USFDA for the treatment of community-acquired pneumonia. ABE-COPD is caused by respiratory tract and pulmonary parenchyma that cause chronic pulmonary inflammation and obstruction in the respiratory tract, which leads to irreversible damage. In the nonclinical evaluation process, zabofloxacin showed strong antibiotic activity on respiratory germs (e.g., Streptococcus pneumonia, S. Haemophilus, S. moraxella) and was the most potent antibacterial agent against penicillin-resistant S. pneumoniae (PRSP) in the murine systemic infection model.
Synthesis
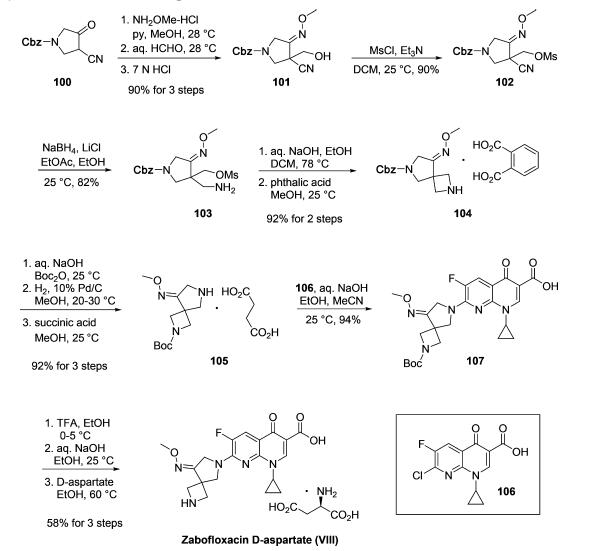
The synthesis of zabofloxacin leverages the wide commercial availability of chloronaphthyridinone acid 106 to essentially reduce the task to the construction of functionalized diazaspirocyclic pyrrolidine 105. As described in a series of patents from researchers at Dong Wha who have exemplified the synthesis on multikilogram scale, the route began with first converting the commercially available ketone 100 to the corresponding oxime followed by formylation to give oximyl alcohol 101. Next, mesylation of the alcohol was followed by conversion of the nitrile to the corresponding amine 103. An intramolecular ring closing step then occurred to secure the azetidine using aqueous sodium hydroxide. Salt formation with phthalic acid furnished 104 in good yield. Next, Boc-protection of the azetidine followed by hydrogenative Cbz removal and treatment with succinic acid resulted in the formation of amine salt 105, and this was followed by a substitution reaction with 106 to deliver the Boc-protected zabofloxacin structure 107. Lastly, removal of Boc via TFA followed by basification and subjection to D-aspartate in warm ethanol furnished zabofloxacin D-aspartate (VIII) in 56% yield for the three-step sequence.
Properties of Zabofloxacin
| storage temp. | Store at -20°C |
| solubility | Soluble in DMSO |
| form | Solid |
| color | White to off-white |
Safety information for Zabofloxacin
Computed Descriptors for Zabofloxacin
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloropyridazine 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 6-Methyl-2-pyridinemethanol 2-Pyridinecarboxaldehdye tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 1-Indanone 2-Chloromethyl-6-methyl-pyridine 4-Morpholinoaniline Abiraterone Ethyl Ether 1-nitro-3,5-dimethyl adamantine (NMEM) Vardenafil Bis-sulphonamide(Dimer) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneYou may like
-
![3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
2304754-51-4 -
 1126-74-5 99%+View Details
1126-74-5 99%+View Details
1126-74-5 -
 2-Fluoro-6-iodobenzoic acid 99%+View Details
2-Fluoro-6-iodobenzoic acid 99%+View Details
111771-08-5 -
 120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 -
 2-pyridineacetonitrile 2739-97-1 99%+View Details
2-pyridineacetonitrile 2739-97-1 99%+View Details
2739-97-1 -
 4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
138163-08-3 -
![ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
313544-31-9 -
 1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
91419-52-2
