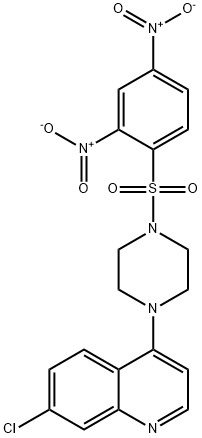
VR23
- CAS NO.:1624602-30-7
- Empirical Formula: C19H16ClN5O6S
- Molecular Weight: 477.88
- MDL number: MFCD28502093
- Update Date: 2026-05-11 19:39:08
What is VR23?
Description
VR23 is a chloroquine derivative that functions as a proteasome inhibitor (IC50s = 1 nM, 50-100 nM, and 3 μM for trypsin-like, chymotrypsin-like, and caspase-like proteasomes, respectively). It also deregulates the activity of cyclin E and other centrosomal proteins, resulting in the induction of multiple centrosome amplification, abnormal spindle formation, uneven cytokinesis, irreversible mitotic arrest, and eventually apoptosis that is specific to cancer cells. In combination with the chymotrypsin-like proteasome inhibitor bortezomib , VR23 can produce a synergistic effect in killing multiple myeloma cells, including those that are resistant to bortezomib.
The Uses of VR23
VR23 is a quinoline-sulfonyl hybrid proteasome inhibitor which is used in cancer therapeutics. Targets cancer via cycline E-mediated centrosome.
in vivo
vr23 was effective in controlling metastatic breast cancer cells and multiple myelomas [1]. in engrafted animals, the treatment with vr23 at 30 mg/kg twice per week for three weeks inhibited tumor growth effectively. following 24 hours pre-treatment with paclitaxel at 20mg/kg/week, the administration of vr23 enhanced the anti-tumor activity of paclitaxel by 70%. histological data showed that vr23 substantially decreased mitotic index, inhibited tumor infiltration into surrounding tissues and remarkably reduced tumor cell proliferation and angiogenesis [4].
References
[1]. pundir s, vu hy, solomon vr, et al. vr23: a quinoline-sulfonyl hybrid proteasome inhibitor that selectively kills cancer via cyclin e-mediated centrosome amplification. cancer research, 2015, 75(19): 4164-4175.
[2]. almond jb, cohen gm. the proteasome: a novel target for cancer chemotherapy. leukemia, 2002, 16(4): 433-443.
[3]. adams j, palombella vj, sausville ea, et al. proteasome inhibitors: a novel class of potent and effective antitumor agents. cancer research, 1999, 59(11): 2615-2622.
[4]. vu hyt, pundir s, solomon rv, et al. anticancer effects and mechanism of vr23, a novel chloroquine derivative. cancer research, 2014, 74(19 supplement): 4545-4545.
Properties of VR23
| storage temp. | Store at -20°C |
| solubility | insoluble in EtOH; insoluble in H2O; ≥45.55 mg/mL in DMSO |
| form | Powder |
| color | Light yellow to yellow |
Safety information for VR23
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for VR23
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran
You may like
-
 Vr23 95% CAS 1624602-30-7View Details
Vr23 95% CAS 1624602-30-7View Details
1624602-30-7 -
 VR23 CAS 1624602-30-7View Details
VR23 CAS 1624602-30-7View Details
1624602-30-7 -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1