Umbralisib
- CAS NO.:1532533-67-7
- Empirical Formula: C31H24F3N5O3
- Molecular Weight: 571.55
- MDL number: MFCD28386165
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-21 10:31:43
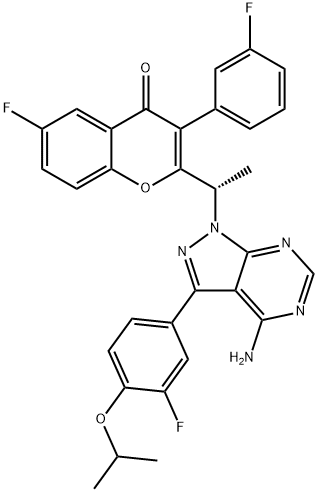
What is Umbralisib?
Absorption
Umbralisib is rapidly absorbed in the GI tract. The Tmax of umbralisib is about 4 hours. After consumption of a high-fat, high calorie meal with umbralisib, the AUC increased by 61% and the Cmax increased by 115%.
Toxicity
The oral LD50 of umbralisib in rats is 3320 mg/kg. Overdose information is not readily available on prescribing information or the literature. In any overdose, supportive and symptomatic treatment should be provided as required.
Description
Umbralisib (TGR-1202) tosylate is an orally active, potent and selective dual PI3Kδ and casein kinase-1-ε (CK1ε) inhibitor, with EC50 of 22.2 nM and 6.0 μM, respectively. Umbralisib tosylate exhibits unique immunomodulatory effects on chronic lymphocytic leukemia (CLL) T cells. Umbralisib tosylate can be used for haematological malignancies reseach.
The Uses of Umbralisib
RP 5264 is a novel PI3K inhibitor that enhances Brentuximab Vedotin-induced lymphoma cell death.
Background
Marginal zone lymphoma is a rare, slowly progressing type of non-Hodgkin lymphoma initially treated with rituximab (an anti-CD20 drug), either alone or in combination with chemotherapy. Unfortunately, many patients experience a relapse or develop resistance to these drugs. Treatment options then become limited, and alternate treatments for the lymphoma are required to control disease progression. Follicular lymphoma is also treated with rituximab and other chemotherapeutic agents, but may show similar progression.
On February 5, 2021, the Food and Drug Administration granted accelerated approval to umbralisib, a kinase inhibitor for PI3K-delta and casein kinase CK1-epsilon, based on promising results from clinical trials. It was marketed as Ukoniq by TG Therapeutics and has been approved for the treatment of relapsing and refractory marginal cell lymphoma and follicular lymphoma in adults. Umbralisib inhibits casein kinase, a primary regulator of protein translation, kinase-1ε, distinguishing it from other lymphoma treatments. While it initially offered a promising therapy for patients experiencing relapsing or refractory disease, umbralisib was withdrawn from the market due to safety concerns as the drug was associated with a possible increased risk of death outweighing the benefits.
Indications
Umbralisib does not have any approved therapeutic indications.
brand name
Ukonig
General Description
Class: lipid kinase; Treatment: MZL, FL; Other name: TGR-1202; Elimination half-life = 91 h; Protein binding > 99.7%
Pharmacokinetics
Umbralisib has a prolonged half-life of 91 h, which enables once-daily dosing. However, the recommended dosage of 800 mg is the highest among all the PI3K inhibitors in use, presumably due to its limited oral bioavailability.
Pharmacokinetics
Umbralisib acts against against marginal zone lymphoma by interrupting the PI3K pathway; this is an essential pathway for B-cell receptor signaling responsible for the progression of lymphoma. In addition, Umbralisib inhibits other pathways involved in specific types of lymphoma, including the casein kinase pathway. An overall response rate of 55% was recorded during clinical trials and the rate of 1-year progression free survival from marginal zone lymphoma was 71%.
A relationship between higher umbralisib steady state exposures and higher incidence of adverse reactions, including diarrhea and elevated AST/ALT was observed during clinical studies. The effect of this drug on QT interval has not been fully characterized.
Synthesis
The synthesis of Umbralisib is as follows:
To a solution of intermediate 13 (0.134 g, 0.494 mmol) in THF (2.0 ml), intermediate 5 (0.150 g, 0.494 mmol) and triphenylphosphine (0.194 g, 0.741 mml) were added and stirred at RT for 5 min. Diisopropylazodicarboxylate ( 0.15 ml, 0.749 mmol) was added heated to 45°C. After 2h, the reaction mixture was quenched with with water and extracted with ethyl acetate. The organic layer was dried over sodium sulphate and concentrated under reduced pressure. The crude product was purified by column chromatography with ethyl acetate : petroleum ether to afford Umbralisib as an off-white solid (0.049 g, 20 %).
Metabolism
During in vitro studies, umbralisib was metabolized by CYP2C9, CYP3A4, and CYP1A2 enzymes.
Properties of Umbralisib
| Boiling point: | 737.4±60.0 °C(Predicted) |
| Density | 1.43±0.1 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | DMF: 1 mg/ml; DMSO: 1 mg/ml |
| form | solid |
| pka | 3.79±0.30(Predicted) |
| color | White to off-white |
Safety information for Umbralisib
Computed Descriptors for Umbralisib
New Products
Dexamethasone Sodium Phosphate 2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid 8-Bromo-3-methyl xanthine Maleic hydrazide m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 2-Fluoro-6-iodobenzoic acid 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 1-Boc-4-cyanopiperidine 3-Pyridineacrylic acid Abiraterone Ethyl Ether Vardenafil Bis-sulphonamide(Dimer) 1-nitro-3,5-dimethyl adamantine (NMEM) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5You may like
-
 141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 -
 Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
27143-07-3 -
 94-71-3 98%View Details
94-71-3 98%View Details
94-71-3 -
 Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
33045-52-2 -
 88965-00-8 98%View Details
88965-00-8 98%View Details
88965-00-8 -
 3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
107228-51-3 -
 4640-67-9 98%View Details
4640-67-9 98%View Details
4640-67-9 -
![2-[(2-Ethoxyphenoxy)methyl]oxirane 98%](https://img.chemicalbook.in//Content/image/CP5.jpg) 2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
5296-35-5
