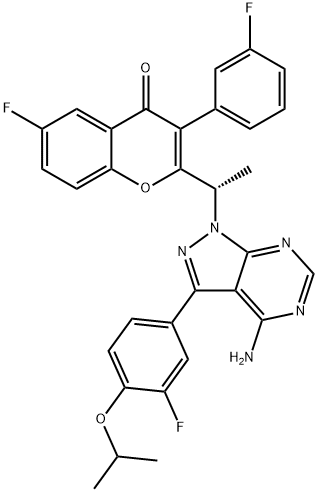
CHEMICAL AND PHYSICAL PROPERTIES
| Melting Point | 139-142 |
|---|---|
| Solubility | ~100 mg/ml |
| LogP | 7.24 |
| Dissociation Constants | 2.71 |
COMPUTED DESCRIPTORS
| Molecular Weight | 571.5 g/mol |
|---|---|
| XLogP3 | 5.8 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 6 |
| Exact Mass | 571.18312413 g/mol |
| Monoisotopic Mass | 571.18312413 g/mol |
| Topological Polar Surface Area | 105 Ų |
| Heavy Atom Count | 42 |
| Formal Charge | 0 |
| Complexity | 1020 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Marginal zone lymphoma is a rare, slowly progressing type of non-Hodgkin lymphoma initially treated with [rituximab] (an anti-CD20 drug), either alone or in combination with chemotherapy. Unfortunately, many patients experience a relapse or develop resistance to these drugs. Treatment options then become limited, and alternate treatments for the lymphoma are required to control disease progression. Follicular lymphoma is also treated with rituximab and other chemotherapeutic agents, but may show similar progression. On February 5, 2021, the Food and Drug Administration granted accelerated approval to umbralisib, a kinase inhibitor for PI3K-delta and casein kinase CK1-epsilon, based on promising results from clinical trials. It was marketed as Ukoniq by TG Therapeutics and has been approved for the treatment of relapsing and refractory marginal cell lymphoma and follicular lymphoma in adults. Umbralisib inhibits casein kinase, a primary regulator of protein translation, kinase-1ε, distinguishing it from other lymphoma treatments. While it initially offered a promising therapy for patients experiencing relapsing or refractory disease, umbralisib was withdrawn from the market due to safety concerns as the drug was associated with a possible increased risk of death outweighing the benefits.