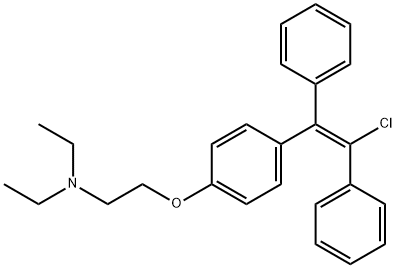
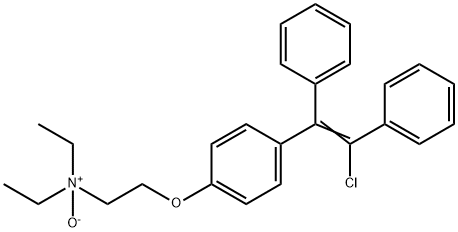
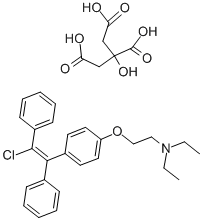
Enclomiphene
- CAS NO.:15690-57-0
- Empirical Formula: C26H28ClNO
- Molecular Weight: 405.96
- MDL number: MFCD08060098
- EINECS: 213-008-6
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 14:58:18
What is Enclomiphene?
Description
Enclomiphene is a synthetic and nonsteroidal antiestrogen. It is the more potent isomer and the one responsible for its ovulation-inducing actions. The half-life of enclomiphene is relatively short, so serum concentrations rise and fall quickly during and after treatment. Antiestrogens represent the first-line treatment strategy in WHO class-2 anovulatory infertility.
The Uses of Enclomiphene
trans-Clomiphene is a Clomiphene (C587025) stereoisomer, a synthetic estrogen agonist-antagonist. Gonad-stimulating principle. Treatment for infertility.
Definition
ChEBI: Clomiphene is a tertiary amine. It has a role as an estrogen antagonist and an estrogen receptor modulator. It derives from a hydride of a stilbene.
Preparation
CONDENSATION OF 4-HYDROXYBENZOPHENONE WITH 2-(DIETHYLAMINO)ETHYL CHLORIDE FOLLOWED BY TREATMENT WITH BENZYL MAGNESIUM CHLORIDE AND DEHYDRATION TO GIVE 2-(P-(1,2-DIPHENYLVINYL)PHENOXY)TRIETHYLAMINE WHICH IS CHLORINATED AND TREATED WITH CITRIC ACID /CITRATE/
Biological Activity
Enclomiphene is the trans-isomer of clomiphene citrate (CC). It has a higher rate of clearance and is less active than the cis-isomer, cis-clomiphene. Clomiphene citrate has been evaluated for antineoplastic activity against breast cancer.
Mode of action
Enclomiphene is a non-steroidal estrogen receptor antagonist that promotes gonadotropin-dependent testosterone secretion by the testes. The main mechanism of its action is thought to be its antagonistic effects on the hypothalamic-pituitary axis and stimulation of GnRH secretion.
References
[1] SIMON HILL Richard Q Vijayaraman Arutchelvam. Enclomiphene, an estrogen receptor antagonist for the treatment of testosterone deficiency in men.[J]. Idrugs, 2009, 12 2: 109-119.
[2] KATHERINE M RODRIGUEZ Larry I L Alexander W Pastuszak. Enclomiphene citrate for the treatment of secondary male hypogonadism.[J]. Expert Opinion on Pharmacotherapy, 2016, 17 11: 1561-1567. DOI:10.1080/14656566.2016.1204294.
[3] JOSHUA A EARL Edward D K. Enclomiphene citrate: A treatment that maintains fertility in men with secondary hypogonadism.[J]. Expert Review of Endocrinology & Metabolism, 2019, 14 3: 157-165. DOI:10.1080/17446651.2019.1612239.
Properties of Enclomiphene
| Melting point: | 149.0-150.5° |
| Boiling point: | 509.0±50.0 °C(Predicted) |
| Density | 1.104±0.06 g/cm3(Predicted) |
| pka | 9.60±0.25(Predicted) |
Safety information for Enclomiphene
Computed Descriptors for Enclomiphene
New Products
2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid 8-Bromo-3-methyl xanthine Maleic hydrazide 4-Bromobenzaldehyde 3-Bromobenzaldehyde 2-Chlorobenzaldehyde 3-Fluorobenzaldehyde 3-Amino-3-(3-fluorophenyl)propanoic acid 1-Propyl-4-piperidone 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 2-Fluoro-6-iodobenzoic acid 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 1-Boc-4-cyanopiperidine 3-Pyridineacrylic acid ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate N-Nitroso Nortriptyline Vardenafil Bis-sulphonamide(Dimer) 1-nitro-3,5-dimethyl adamantine (NMEM) Nabumetone Impurity 5 3-(2-aminoethyl) benzene sulfonamide 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran

You may like
-
 trans-Clomiphene 15690-57-0 97%View Details
trans-Clomiphene 15690-57-0 97%View Details
15690-57-0 -
![1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%View Details
1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%View Details
1620885-59-7 -
 (S)-2-Bromobutanoic acid 32659-49-7 97-99%View Details
(S)-2-Bromobutanoic acid 32659-49-7 97-99%View Details
32659-49-7 -
 Deuterated Styrene-d8 97-99%View Details
Deuterated Styrene-d8 97-99%View Details
19361-62-7 -
![ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 1872090-65-7 Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
1872090-65-7 Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
1872090-65-7 -
 6-Chloro-3,4-dimethylpyridin-2-amine 1503408-77-2 97-99%View Details
6-Chloro-3,4-dimethylpyridin-2-amine 1503408-77-2 97-99%View Details
1503408-77-2 -
 1-(3,5-dichlorophenyl)-2,2,2-trifluoroethan-1-ol 1187989-89-4 97-99%View Details
1-(3,5-dichlorophenyl)-2,2,2-trifluoroethan-1-ol 1187989-89-4 97-99%View Details
1187989-89-4
