SULFURYL CHLORIDE FLUORIDE
Synonym(s):Chlorosulfonyl fluoride;Fluorosulfonyl chloride;Sulfuryl chlorofluoride
- CAS NO.:13637-84-8
- Empirical Formula: ClFO2S
- Molecular Weight: 118.52
- MDL number: MFCD00011607
- EINECS: 237-126-2
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-31 14:38:37
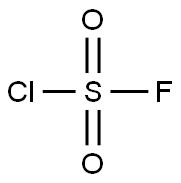
What is SULFURYL CHLORIDE FLUORIDE?
Description
Sulfuryl chloride fluoride (SO2ClF) is a strong Lewis acid and a useful solvent and reagent. As shown in the hazard information table, it must be handled with care. Its boiling point is in the range that allows its use as a gas or a liquid.
In the 1930s, Harold Simmons Booth* and Carl V. Herrmann at Western Reserve University (Cleveland; later Case Western Reserve University) performed pioneering work on SO2ClF. They synthesized it via the reaction of sulfuryl chloride (SO2Cl2) and antimony trifluoride (SbF3) in the presence of antimony pentachloride (SbCl5). They also measured several properties of SO2ClF, including boiling point, melting point, liquid density, vapor pressure from –89 to +9 oC, heat of vaporization, and surface tension.
But the real champion of SO2ClF was the 1994 chemistry Nobelist George A. Olah. In 1967, also at Case Western, Olah and Joachim Lukas used the solvent system SO2ClF–fluorosulfonic acid (FSO3H)–antimony pentafluoride (SbF5) to generate stable alkylcarbonium ions (now called carbocations)—from simple ones such as Me3C+ and Me2EtC+ through cations containing as many as nine carbon atoms. Acid mixtures like the ones they used came to be known as superacids.
In 1976, Olah, Mark R. Bruce, and John Welch developed an improved synthesis of SO2ClF by adding SO2Cl2 to pyridinium poly(hydrogen fluoride) under mild conditions. By the early 1980s, now at the University of Southern California (Los Angeles), Olah and co-workers developed SO2ClF as a versatile reagent. Some SO2ClF applications include
Finally, in 1992, V. Prakash Reddy, Donald R. Bellew, and G. K. Surya Prakash* at USC reported another useful synthesis of SO2ClF, from SO2Cl2 and ammonium or potassium fluoride in the presence of trifluoroacetic acid. Surya Prakash was a student of Olah and is his successor as the director of USC’s Loker Hydrocarbon Research Institute, the location of much of Olah’s carbocation research.
Chemical properties
clear colorless to light yellow liquid
Definition
ChEBI: Sulfuryl chloride fluoride is a sulfuryl halide.
Safety Profile
Poison by inhalation. When heated to decomposition it emits very toxic fumes of Cl-, F-, and SOx. See also FLUORIDES, CHLORIDES, and SULFIDES.
Properties of SULFURYL CHLORIDE FLUORIDE
| Melting point: | -125 °C |
| Boiling point: | 7 °C |
| Density | 1.623 g/cm3(Temp: 0 °C) |
| Flash point: | 113 °C |
| storage temp. | -20°C |
| solubility | reacts |
| solubility | reacts with H2O |
| form | gas |
| appearance | colorless gas or colorless to light yellow liquid |
| color | colorless |
| Sensitive | Moisture Sensitive |
| CAS DataBase Reference | 13637-84-8(CAS DataBase Reference) |
| EPA Substance Registry System | Sulfuryl chloride fluoride (13637-84-8) |
Safety information for SULFURYL CHLORIDE FLUORIDE
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Skull and Crossbones Acute Toxicity GHS06 |
| GHS Hazard Statements |
H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P260:Do not breathe dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. |
Computed Descriptors for SULFURYL CHLORIDE FLUORIDE
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate 3,6-Dichloropyridazine Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane 4-Morpholinoaniline 2-Chloromethyl-6-methyl-pyridine 1-Indanone tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 2-Pyridinecarboxaldehdye 6-Methyl-2-pyridinemethanol 4-Bromo-2-fluoro-N-methylbenzamide 3-Azetidinecarboxylic acid N-(2-(4-((4-Fluorobenzyl)carbamoyl)-5-hydroxy-6-methoxypyrimidin-2-yl)propan-2-yl)-5-methyl-1,3,4-oxadiazole-2-carboxamide (R)-4-Boc-morpholine-3-carboxylic acid (Required high chiral purity NLT:99.5%) Tetrahydro-3-(2-methyl-2- nitropropyl)-2H-pyran-2-one 3-Bromo-2-fluorobenzonitrile Desoximetasone Boron Triiodide Nicotinic acid RAPAMYCIN Pentachlorobenzonitrile BudesonideRelated products of tetrahydrofuran
You may like
-
 1589503?95?6 2-((2,6-Difluorobenzyl)(ethoxycarbonyl)amino)-4-((dimethylamino)methyl)-5-(4-nitrophenyl)thiophene-3-carboxylic acid 99%View Details
1589503?95?6 2-((2,6-Difluorobenzyl)(ethoxycarbonyl)amino)-4-((dimethylamino)methyl)-5-(4-nitrophenyl)thiophene-3-carboxylic acid 99%View Details
1589503?95?6 -
 2-Chlorophenyl diphenyl chloromethane 99%View Details
2-Chlorophenyl diphenyl chloromethane 99%View Details
42074-68-0 -
 6-(chloromethyl)pteridine-2,4-diamine monohydrochloride 82778?08?3 99%View Details
6-(chloromethyl)pteridine-2,4-diamine monohydrochloride 82778?08?3 99%View Details
82778?08?3 -
 Ethyl (Z)-3-(tetrahydro-2H-pyran-4-yl)acrylate 1373168-67-2 99%View Details
Ethyl (Z)-3-(tetrahydro-2H-pyran-4-yl)acrylate 1373168-67-2 99%View Details
1373168-67-2 -
 1,3-Dibromo-5-fluorobenzene 99%View Details
1,3-Dibromo-5-fluorobenzene 99%View Details
1435-51-2 -
![729559-09-5 [N(E)]-N-Ethylidene-2-methyl-2-propanesulfinamide 99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 729559-09-5 [N(E)]-N-Ethylidene-2-methyl-2-propanesulfinamide 99%View Details
729559-09-5 [N(E)]-N-Ethylidene-2-methyl-2-propanesulfinamide 99%View Details
729559-09-5 -
 4-Isopropyl Thiophenol 4946-14-9 99%View Details
4-Isopropyl Thiophenol 4946-14-9 99%View Details
4946-14-9 -
 2709271-25-8 99%View Details
2709271-25-8 99%View Details
2709271-25-8