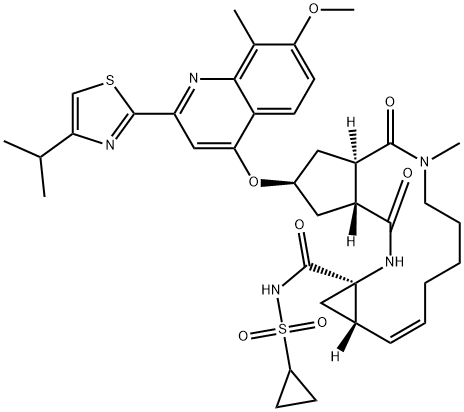
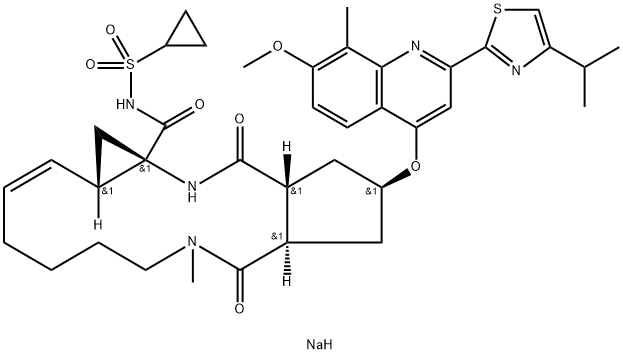
Simeprevir sodium
- CAS NO.:1241946-89-3
- Empirical Formula: C38H47N5O7S2.x(Na)
- Molecular Weight: 773.94
- MDL number: MFCD30724946
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-28 04:12:19
What is Simeprevir sodium?
Description
Simeprevir is an orally bioavailable and potent inhibitor of the hepatitis C virus (HCV) nonstructural protein 3/4A (NS3/4A) protease (Ki = 0.36 nM), a serine protease essential for HCV replication. It inhibits HCV viral replication with an EC50 value of 7.8 nM in Huh7 replicon cells using a luciferase-based assay. Simeprevir is effective against the HCV genotypes 1a and 1b in biochemical assays. In Huh7 replicon cells, it is synergistically effective when used in combination with IFN-α or NM-107 and has an additive effect when used with ribavirin . Formulations containing simeprevir have been used, alone or in combination with pegylated IFN-α and ribavirin, for the treatment of HCV.
Properties of Simeprevir sodium
| storage temp. | Store at -20°C |
| solubility | DMSO:30.0(Max Conc. mg/mL);38.86(Max Conc. mM) DMSO:PBS (pH 7.2) (1:2):0.33(Max Conc. mg/mL);0.43(Max Conc. mM) DMF:30.0(Max Conc. mg/mL);38.86(Max Conc. mM) |
| form | A crystalline solid |
Safety information for Simeprevir sodium
Computed Descriptors for Simeprevir sodium
Simeprevir sodium manufacturer
New Products
Pentadecanoic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 2-Bromo-5-Iodopyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile Tetrahydropyranyl-4-acetic acid Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside 5-fluoro-1,3-benzodioxole 2-Bromo-6-fluoroaniline 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-oneRelated products of tetrahydrofuran
You may like
-
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
Statement: All products displayed on this website are only used for non medical purposes such as industrial applications or scientific research, and cannot be used for clinical diagnosis or treatment of humans or animals. They are not medicinal or edible.