RILPIVIRINE HCL
- CAS NO.:700361-47-3
- Empirical Formula: C22H19ClN6
- Molecular Weight: 402.89
- MDL number: MFCD11046523
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 14:59:02
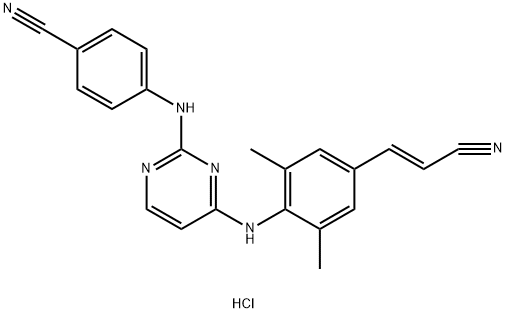
What is RILPIVIRINE HCL?
The Uses of RILPIVIRINE HCL
Rilpivirine Hydrochloride was shown to be able to treat and or prevent immunodeficiency virus-1. It also has uses for anti-viral therapy
Definition
ChEBI: A hydrochloride obtained by reaction of rilpivirine with one equivalent of hydrochloric acid. Used for treatment of HIV.
Clinical Use
Rilpivirine hydrochloride (Edurant), a non-nucleoside reverse transcriptase inhibitor (NNRTI), received its approval both from the U.S. FDA and E.U. EMA in 2011 for the treatment of HIV-1 infection in treatment-na?ve adult patients. It was discovered and developed by Janssen Pharmaceuticals and its subsidiary Tibotec Pharmaceuticals. As a second generation NNRTI, rilpivirine hydrochloride displayed higher potency and longer half-life with a 25 mg once a day dose, compared to existing NNRTIs, such as the 200 mg BID of efavirenze (Sustiva). In late 2011, the fixed-dose combination products of rilpivirine hydrochloride with two nucleoside reverse transcriptase inhibitor (RTIs) emtricitabine and tenofovir disoproxil fumarate, co-developed by Gilead Science and Tibotec, were also approved both by the FDA and EMA under brand names Complera® and Eviplera®, respectively.
Synthesis
Similar to efavirenze, rilpivirine hydrochloride is a diarylpyrimidine (DAPY) compound, and the large-scale process synthesis begins with commercially available 2-methylthio-4-pyrimidinone (193) shown in the scheme.
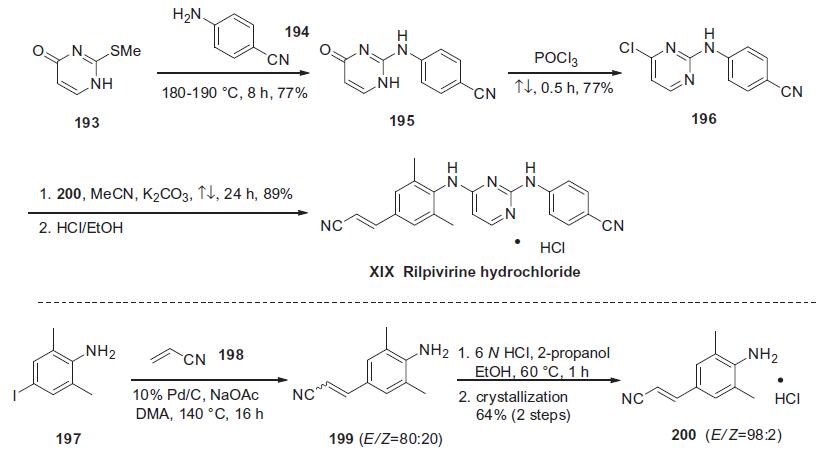
Thioether 193 was condensed with neat 4-cyanoaniline (194) at elevated temperature to afford diarylamine 195 in 77% yield. Subsequent treatment of pyrimidone 195 with refluxing POCl3 provided the corresponding chloride 196 in 77% yield.160,161 In the presence of K2CO3, chloride 196 was treated with the (E)-cinnamonitrile aniline 200 to give rilpivirine hydrochloride (XIX) in good yield.158 Aniline 200 was prepared via a Heck reaction of commercially available 4-iodo-2,6-dimethyl-benzeneamine (197) and acrylonitrile (198) affording compound 199 as a 4:1 mixture of E/Z isomers. The distribution of E/Z olefins was increased to 98:2 by salt formation and recrystallization to ultimately provide pure (E)-200 in 64% yield for two steps.
Properties of RILPIVIRINE HCL
| storage temp. | Store at -20°C |
| solubility | DMSO:50.0(Max Conc. mg/mL);136.46(Max Conc. mM) |
| form | Solid |
| color | White to off-white |
Safety information for RILPIVIRINE HCL
Computed Descriptors for RILPIVIRINE HCL
New Products
Pentadecanoic acid 3-Bromophenylacetic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-Amino-5-cyanopyridine 2-Bromo-5-cyanopyridine 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 3′-Hydroxyacetophenone 5-fluoro-1,3-benzodioxole 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 2-Bromo-6-fluoroaniline 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole N-(4-Bromophenyl)-2-chloroacetamide 6-Isopropylpicolinonitrile N-Methyl-3,5-dinitro-2-pyridinamine Phenyl-boronic acid-d5 10,11-Dihdyro-10-oxo-5H-dibenz[b,f]azepine-5-carboxamide-d4 5-(2,4-difluorophenyl)oxazolidin-2-oneRelated products of tetrahydrofuran
![4-[[4-[[4-[(E)-2-cyanoethenyl]-2,6-dimethyl-phenyl]amino]pyrimidin-2-yl]amino]benzonitrile](https://img.chemicalbook.in/CAS/GIF/500287-72-9.gif)
![DAPIVIRINE,4-[[4-(2,4,6-TRIMETHYLPHENYL)AMINO]PYRIMIDIN-2-YL]AMINO]BENZONITRILE](https://img.chemicalbook.in/CAS/GIF/244767-67-7.gif)

You may like
-
 700361-47-3 Rilpivirine hydrochloride 98%View Details
700361-47-3 Rilpivirine hydrochloride 98%View Details
700361-47-3 -
 700361-47-3 99%View Details
700361-47-3 99%View Details
700361-47-3 -
 Rilpivirine hydrochloride 99%View Details
Rilpivirine hydrochloride 99%View Details
700361-47-3 -
 700361-47-3 95-99%View Details
700361-47-3 95-99%View Details
700361-47-3 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
1872090-65-7 -
 32659-49-7 97-99%View Details
32659-49-7 97-99%View Details
32659-49-7
