madopar
- CAS NO.:37270-69-2
- Empirical Formula: C10H15N3O5.C9H11NO4
- Molecular Weight: 454.433
- SAFETY DATA SHEET (SDS)
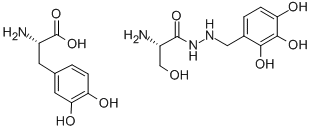
What is madopar?
Drug interactions
Potentially hazardous interactions with other drugs
Anaesthetics: risk of arrhythmias with volatile liquid
anaesthetics such as halothane
.
Antidepressants: hypertensive crisis with MAOIs
and linezolid (including moclobemide) - avoid for at
least 2 weeks after stopping MAOI.
Bupropion: increased risk of side effects of levodopa
Ferrous sulphate: reduces AUC of levodopa by
30-50%, clinically significant in some but not all
patients.
Metabolism
Levodopa is rapidly decarboxylated by the enzyme
aromatic l-amino acid decarboxylase, mostly in the gut,
liver, and kidney, to dopamine, which is metabolised
in turn, principally to dihydroxyphenylacetic acid
(DOPAC) and homovanillic acid (HVA). Other routes
of metabolism include O-methylation, transamination,
and oxidation, producing a variety of minor metabolites
including noradrenaline and 3-O-methyldopa; the latter
may accumulate in the CNS due to its relatively long
half-life.
About 80% of an oral dose of levodopa is excreted in the
urine within 24 hours, mainly as dihydroxyphenylacetic
and homovanillic acids. Only small amounts of levodopa
are excreted unchanged in the faeces.
Benserazide is rapidly excreted in the urine in the form
of metabolites, mostly within the first 6 hours; 85% of
urinary excretion occurrs within 12 hours. It is mainly
metabolised in the gut and appears to protect levodopa
against decarboxylation primarily in the gut, but alsoin the rest of the body, mainly by way of its metabolite
trihydroxybenzylhydrazine.
Safety information for madopar
New Products
2-(2-aminoethyl) benzenesulfonamide Gemcitabine 3-benzoate impurity Dihydroxyphenylacetone Ornidazole EP impurity D Acetyl amino diol impurity Valsartan Benzyl Ester Impurity B N,N'-Dicyclohexylcarbodiimide 3-Bromo-2-Methyl-5-Nitropyridine 5-Bromo-3-Methyl-2-Pyridinecarboxylic acid 2-Amino-4-Methylpyridine 4-Aminopyridine 4-dimethylaminopyridineRelated products of tetrahydrofuran

You may like
-
 4-(Methylnitrosoamino)-butanal NLT 95%View Details
4-(Methylnitrosoamino)-butanal NLT 95%View Details
339362-86-6 -
 848696-99-1 NLT 95%View Details
848696-99-1 NLT 95%View Details
848696-99-1 -
 Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
1384752-15-1 -
 615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 -
 175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 -
 S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
1346605-15-9 -
 Valsartan Related Compound A NLT 95%View Details
Valsartan Related Compound A NLT 95%View Details
137862-87-4 -
 Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
124750-49-8
