LOVASTATIN HYDROXY ACID, SODIUM SALT
- CAS NO.:75225-51-3
- Empirical Formula: C24H38O6
- Molecular Weight: 422.55
- MDL number: MFCD03425588
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-04-23 13:52:06
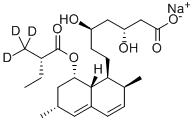
What is LOVASTATIN HYDROXY ACID, SODIUM SALT?
The Uses of LOVASTATIN HYDROXY ACID, SODIUM SALT
Lovastatin Hydroxy Acid, Sodium Salt (Lovastatin EP Impurity B) is a metabolite of Lovastatin.
Definition
ChEBI: A polyketide obtained by hydrolysis of the pyranone ring of lovastatin.
Properties of LOVASTATIN HYDROXY ACID, SODIUM SALT
| Melting point: | 140 - 143oC |
| Boiling point: | 602.3±55.0 °C(Predicted) |
| Density | 1.14±0.1 g/cm3(Predicted) |
| storage temp. | Hygroscopic, -20°C Freezer, Under Inert Atmosphere |
| solubility | DMSO (Slightly), Methanol (Slightly, Sonicated), Water (Slightly) |
| form | Solid |
| pka | 4.31±0.10(Predicted) |
| color | Off-White to Light Brown |
| Stability: | Very Hygroscopic |
Safety information for LOVASTATIN HYDROXY ACID, SODIUM SALT
| Signal word | Warning |
| Pictogram(s) |
 Health Hazard GHS08 |
| GHS Hazard Statements |
H351:Carcinogenicity H413:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P202:Do not handle until all safety precautions have been read and understood. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P308+P313:IF exposed or concerned: Get medical advice/attention. P405:Store locked up. P501:Dispose of contents/container to..… |
Computed Descriptors for LOVASTATIN HYDROXY ACID, SODIUM SALT
New Products
2-(2-aminoethyl) benzenesulfonamide Gemcitabine 3-benzoate impurity Dihydroxyphenylacetone Ornidazole EP impurity D Acetyl amino diol impurity Valsartan Benzyl Ester Impurity B N,N'-Dicyclohexylcarbodiimide 3-Bromo-2-Methyl-5-Nitropyridine 5-Bromo-3-Methyl-2-Pyridinecarboxylic acid 2-Amino-4-Methylpyridine 4-Aminopyridine 4-dimethylaminopyridineRelated products of tetrahydrofuran


![SIMVASTATIN ACID, SODIUM SALT [3H(G)]](https://img.chemicalbook.in/)



![LOVASTATIN ACID [3H(G)] SODIUM SALT](https://img.chemicalbook.in/StructureFile/ChemBookStructure8/GIF/CB6193357.gif)
You may like
-
 4-(Methylnitrosoamino)-butanal NLT 95%View Details
4-(Methylnitrosoamino)-butanal NLT 95%View Details
339362-86-6 -
 848696-99-1 NLT 95%View Details
848696-99-1 NLT 95%View Details
848696-99-1 -
 Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
1384752-15-1 -
 615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 -
 175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 -
 S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
1346605-15-9 -
 Valsartan Related Compound A NLT 95%View Details
Valsartan Related Compound A NLT 95%View Details
137862-87-4 -
 Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
124750-49-8
Statement: All products displayed on this website are only used for non medical purposes such as industrial applications or scientific research, and cannot be used for clinical diagnosis or treatment of humans or animals. They are not medicinal or edible.
