LEAD CYANATE
- CAS NO.:592-05-2
- Empirical Formula: C2N2Pb
- Molecular Weight: 259.2348
- EINECS: 209-742-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-01 16:59:24
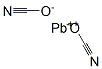
What is LEAD CYANATE?
Chemical properties
white to yellowish powder(s); used in metallurgy [HAW93]
General Description
A white to yellowish powder. Toxic by skin absorption, by ingestion, and by inhalation of dust, or hydrogen cyanide produced by slight decomposition. Produces toxic oxides of nitrogen in fires.
Air & Water Reactions
Slightly soluble in water and gradually decomposed by water evolving poisonous and flammable hydrogen cyanide.
Reactivity Profile
LEAD CYANATE is decomposed by acids to give off hydrogen cyanide, a flammable poisonous gas. Tends to explosive instability. Capable of violent oxidation: fusion with metal chlorates, perchlorates, nitrates or nitrites can cause explosions [Bretherick, 1979 p. 101]. Reacts with incandescence with magnesium [Mellor, 1940, Vol. 4, 271].
Health Hazard
Highly toxic, may be fatal if inhaled, swallowed or absorbed through skin. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Containers may explode when heated. Runoff may pollute waterways.
Properties of LEAD CYANATE
| solubility | slightly soluble in H2O; reacts with acid solutions |
| form | white-yellow powder |
| color | white-yellow |
| Water Solubility | slightly soluble H2O; decomposed by acid [HAW93] |
| EPA Substance Registry System | Lead cyanide (Pb(CN)2) (592-05-2) |
Safety information for LEAD CYANATE
Computed Descriptors for LEAD CYANATE
New Products
Dexamethasone Sodium Phosphate 2-(2-Ethoxyphenoxy)ethyl bromide 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 2-Fluoro-6-iodobenzoic acid 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 1-Boc-4-cyanopiperidine 3-Pyridineacrylic acid Abiraterone Ethyl Ether Vardenafil Bis-sulphonamide(Dimer) 1-nitro-3,5-dimethyl adamantine (NMEM) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 2-Amino-3,5-Dichloro-4-picoline 5-Amino-2-EthoxypyridineRelated products of tetrahydrofuran
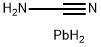
You may like
-
 141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 -
 Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
27143-07-3 -
 94-71-3 98%View Details
94-71-3 98%View Details
94-71-3 -
 Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
33045-52-2 -
 88965-00-8 98%View Details
88965-00-8 98%View Details
88965-00-8 -
 3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
107228-51-3 -
 4640-67-9 98%View Details
4640-67-9 98%View Details
4640-67-9 -
![2-[(2-Ethoxyphenoxy)methyl]oxirane 98%](https://img.chemicalbook.in//Content/image/CP5.jpg) 2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
5296-35-5
