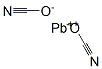CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Lead cyanide appears as a white to yellowish powder. Toxic by skin absorption, by ingestion, and by inhalation of dust, or hydrogen cyanide produced by slight decomposition. Produces toxic oxides of nitrogen in fires. |
|---|---|
| Chemical Classes | Toxic Gases & Vapors -> Cyanides |
COMPUTED DESCRIPTORS
| Molecular Weight | 259 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 259.98280 g/mol |
| Monoisotopic Mass | 259.98280 g/mol |
| Topological Polar Surface Area | 47.6 Ų |
| Heavy Atom Count | 5 |
| Formal Charge | 0 |
| Complexity | 85.3 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Lead cyanide appears as a white to yellowish powder. Toxic by skin absorption, by ingestion, and by inhalation of dust, or hydrogen cyanide produced by slight decomposition. Produces toxic oxides of nitrogen in fires.