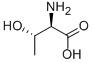
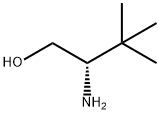
L-Serine
Synonym(s):L-Serine;Ser;L-Serine - CAS 56-45-1 - Calbiochem;(S)-2-Amino-3-hydroxypropionic acid;(S)-(+)-Serine
- CAS NO.:56-45-1
- Empirical Formula: C3H7NO3
- Molecular Weight: 105.09
- MDL number: MFCD00064224
- EINECS: 200-274-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-28 01:22:29
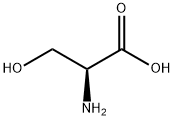
What is L-Serine?
Description
Serine (abbreviated as Ser or S) is an amino acid with the formula HO2CCH(NH2)CH2OH. It is one of the proteinogenic amino acids. Its codons in the genetic code are UCU, UCC, UCA, UCG, AGU and AGC. By virtue of the hydroxyl group, serine is classified as a polar amino acid.
L-serine is an amino acid essential for the synthesis of phosphatidylserine, which is a component of the membrane of brain cells (i.e., neurons). It can be produced in the body, including the brain, but an external supply from the diet is essential in maintaining necessary levels. Although preclinical studies suggest L-serine may inhibit inflammation in the brain, levels of L-serine in humans do not appear to be associated with dementia or cognitive decline. Because L-serine is a naturally occurring amino acid, supplementation is likely safe in moderation.
Description
L-Alanine1 and L-serine1 are amino acids that are necessary for the biosynthesis of proteins. They are considered “nonessential” in human diets (and those of most other animals) because they are synthesized in the body.
In 1850, long before it was discovered in and isolated from natural substances, alanine was synthesized from acetaldehyde by German chemist Adolph Strecker. Of course, Strecker made the D,L-racemate of the amino acid rather than either isomer alone.
Serine, on the other hand, was first discovered in nature, specifically in silk protein, by German chemist Emil Cramer in 1865. And, speaking of silk . . .
Alanine and serine, along with previous Molecules of the Week glycine and L-proline, are the major amino acids in spider silk proteins. Alanine-rich crystalline areas give silk strength, whereas glycine-rich amorphous regions provide elasticity.
Halloween reminds us of spiders. Spiders spin webs of silk. Silk is made up of proteins. Proteins are polymers of amino acids. Who would have thought that such simple molecules could be scary?
1All amino acids that are biosynthesized into proteins are the L-enantiomers. Some D-enantiomers appear in nature, but they are relatively rare.
Chemical properties
L-Serine is a white crystal or crystalline powder, slightly sweet taste, soluble in water and acid, insoluble in alcohol and ether. From soybeans, wine fermentation agent, dairy products, eggs, fish, lactalbumin, meat, nuts, seafood, whey, and whole grains to get. Serine is also used as a dietary supplement where it can aid improved memory function and in personal care products where it helps with the production of new skin cells.
The Uses of L-Serine
L-Serine has been used in the preparation of Tris-BSAN buffer for homogenization. It has also been used for quantitative analysis of excretion of polypeptides in normal urine.
The Uses of L-Serine
L-Serine is used in the synthesis of purines and pyrimidines as antibacterial/antifungal agents, as well as acting as a proteinogenic compound.
The Uses of L-Serine
L-Serine is classified as a non-essential amino acid. It is used as a cell culture media component and raw material for the commercial biomanufacture of therapeutic recombinant proteins and monoclonal antibodies.
What are the applications of Application
L-Serine is L-Serine is a D-amino acid synthesis reagent
Definition
ChEBI: L-serine is the L-enantiomer of serine. It has a role as a human metabolite, an algal metabolite, a Saccharomyces cerevisiae metabolite, an Escherichia coli metabolite and a mouse metabolite. It is a serine family amino acid, a proteinogenic amino acid, a L-alpha-amino acid and a serine. It is a conjugate base of a L-serinium. It is a conjugate acid of a L-serinate. It is an enantiomer of a D-serine. It is a tautomer of a L-serine zwitterion.
Production Methods
Industrially , L - serine is produced by fermentation, with an estimated 100 - 1000 tonnes per year produced . In the laboratory, racemic serine can be prepared from methyl acrylate via several steps.
Biotechnological Production
Serine is the first amino acid produced in the 3-phosphoglycerate pathway. It is further converted to glycine and L-cysteine. Industrially L-serine can be produced by direct fermentation or by an enzymatic process from glycine. The enzymatic route developed by Mitsui reacts glycine with formaldehyde using serine hydroxymethyltransferase (SHMT). With an overexpression of SHMT in E. coli, concentrations of serine of over 300 g/L in 35 h reaction time have been reported, with a glycine conversion of[98 %. This process requires the addition of tetrahydrofolic acid to the system as a cofactor. An alternative to enzymatic production is a direct fermentation to give L-serine. Strains based on Brevibacterium flavum and C. glutamicum have been described. In both strains, the enzymes phosphoglycerate dehydrogenase (serA), phosphoserine phosphatase (serB), and phosphoserine transaminase (serC) have been overexpressed. These enzymes are involved in the biosynthesis pathway from 3-phosphoglycerate. Note that because of product inhibition by L-serine of serA, feedback-resistant mutants have been developed to increase yields. A mutant strain of B. flavum with a feedback-resistant serA* and overexpression of the serA*, serB, and serC has been reported to accumulate 35.2 g/L L-serine with a carbon yield of 32 % based on glucose. In addition it has been shown that increased yields in C. glutamicum can be obtained by deleting the L-serine degrading enzyme L-serine dehydratase (sdaA).
General Description
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Biological Activity
L-Serine is an endogenous agonist at the inhibitory glycine receptor.
L-Serine is a non-essential amino acid that acts as a precursor for nucleotide synthesis. It has roles in the development and function of the central nervous system. L-Serine also has a role in cellular proliferation.
Precursor of glycine by serine hydroxymethyltransferase.
Biochem/physiol Actions
Precursor of glycine by serine hydroxymethyltransferase.
Biotechnological Applications
Metabolic
Serine is important in metabolism in that it participates in the biosynthesis of purines and pyrimidines. It is the precursor to several amino acids including glycine and cysteine, and tryptophan in bacteria. It is also the precursor to numerous other metabolites, including sphingolipids and folate, which is the principal donor of one-carbon fragments in biosynthesis.
Structural role
Serine plays an important role in the catalytic function of many enzymes. It has been shown to occur in the active sites of chymotrypsin, trypsin, and many other enzymes. The so-called nerve gases and many substances used in insecticides have been shown to act by combining with a residue of serine in the active site of acetylcholine esterase, inhibiting the enzyme completely.
As a constituent (residue) of proteins, its side chain can undergo O-linked glycosylation, which may be functionally related to diabetes.
It is one of three amino acid residues that are commonly phosphorylated by kinases during cell signaling in eukaryotes. Phosphorylated serine residues are often referred to as phosphoserine.
Signaling
D-Serine, synthesized in the brain by serine racemase from Lserine (its enantiomer), serves as both a neuro transmitter and a gliotransmitter by coactivating NMDA receptors, making them able to open if they then also bind glutamate. D-serine is a potent agonist at the glycine site of the NMDA-type glutamate receptor. For the receptor to open, glutamate and either glycine or D-serine must bind to it. In fact, D-serine is a more potent agonist at the glycine site on the NMDAR than glycine itself. D-serine was only thought to exist in bacteria until relatively recently; it was the second D amino acid discovered to naturally exist in humans, present as a signalling molecule in the brain, soon after the discovery of D-aspartate. Had D amino acids been discovered in humans sooner, the glycine site on the NMDA receptor might instead be named the D-serine site.
Gustatory sensation
Pure D-Serine is an off-white crystalline powder with a very faint funky or dirty aroma. L-Serine is sweet with minor umami and sour tastes at high concentration. D-Serine is sweet with an additional minor sour taste at medium and high concentrations.
Safety
L-serine is generally safe to take as an oral dietary supplement. In a phase 1 safety trial in 20 ALS patients, L-serine treatment (0.5-15.0 g, twice daily) for 6 months was generally well-tolerated and appeared to be safe. Two patients withdrew from the trial due to gastrointestinal side effects but no other adverse events or changes in routine blood tests measures were observed.
Synthesis
This compound is one of the naturally occurring proteinogenic amino acids. Only the L-stereoisomer appears naturally in proteins. It is not essential to the human diet, since it is synthesized in the body from other metabolites, including glycine. Serine was first obtained from silk protein, a particularly rich source, in 1865. Its name is derived from the Latin for silk, sericum. Serine's structure was established in 1902.
The biosynthesis of serine starts with the oxidation of 3- phosphoglycerate to 3-phosphohydroxypyruvate and NADH. Reductive amination of this ketone followed by hydrolysis gives serine. Serine hydroxymethyltransferase catalyzes the reversible, simultaneous conversions of L-serine to glycine (retro-aldol cleavage) and 5,6,7,8-tetra hydrofolate to 5,10-methylene tetra hydrofolate (hydrolysis).
This compound may also be naturally produced when UV light illuminates simple ices such as a combination of water, methanol, hydrogen cyanide, and ammonia, suggesting that it may be easily produced in cold regions of space.
Purification Methods
A likely impurity is glycine. Crystallise L-serine from H2O by adding 4volumes of EtOH. Dry and store it in a desiccator. It sublimes at 160-170o/0.3mm with 99.7% recovery, and unracemised [Gross & Gradsky J Am
Properties of L-Serine
| Melting point: | 222 °C (dec.) (lit.) |
| Boiling point: | 197.09°C (rough estimate) |
| alpha | 15.2 º (c=10, 2N HCl) |
| Density | 1.6 |
| refractive index | 1.4368 (estimate) |
| Flash point: | 150°C |
| storage temp. | 2-8°C |
| solubility | H2O: 50 mg/mL |
| form | powder |
| appearance | white crystals or powder |
| pka | 2.19(at 25℃) |
| color | White |
| PH | 5-6 (100g/l, H2O, 20℃) |
| Odor | wh. cryst. or cryst. powd., odorless, sweet taste |
| optical activity | [α]20/D +13.5±0.5°, c = 5% in 5 M HCl |
| Water Solubility | 250 g/L (20 ºC) |
| λmax | λ: 260 nm Amax: 0.05 λ: 280 nm Amax: 0.05 |
| Merck | 14,8460 |
| Sublimation | 150 ºC |
| BRN | 1721404 |
| Stability: | Stable. Incompatible with strong oxidizing agents. |
| CAS DataBase Reference | 56-45-1(CAS DataBase Reference) |
| NIST Chemistry Reference | L-Serine(56-45-1) |
| EPA Substance Registry System | L-Serine (56-45-1) |
Safety information for L-Serine
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P302+P352:IF ON SKIN: wash with plenty of soap and water. P304+P340:IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for L-Serine
| InChIKey | MTCFGRXMJLQNBG-REOHCLBHSA-N |
L-Serine manufacturer
Clickchem Research LLP
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 2-Bromo-5-Iodopyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 2-Hydroxy-5-methoxybenzoic acid 2-Cyanopyrimidine Xanthene-9-carboxylic acid 4,4-Diethoxybutanenitrile Methyl 4-amino-2,3-difluoro-5-nitrobenzoate 5-fluoro-1,3-benzodioxole 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 4-Chloro-2-methyl quinoline 2-Amino-4-phenyl-thiazole 2-(Chloromethyl) quinazolin-4(3H)-oneRelated products of tetrahydrofuran
You may like
-
 L-Serine 99%View Details
L-Serine 99%View Details -
 L-Serine for cell culture CAS 56-45-1View Details
L-Serine for cell culture CAS 56-45-1View Details
56-45-1 -
 L-Serine extrapure CHR CAS 56-45-1View Details
L-Serine extrapure CHR CAS 56-45-1View Details
56-45-1 -
 L-Serine for tissue culture CAS 56-45-1View Details
L-Serine for tissue culture CAS 56-45-1View Details
56-45-1 -
 L-Serine ExiPlus CAS 56-45-1View Details
L-Serine ExiPlus CAS 56-45-1View Details
56-45-1 -
 L-Serine 99% CASView Details
L-Serine 99% CASView Details -
 L-SERINE 90 % AboveView Details
L-SERINE 90 % AboveView Details -
 L-SerineView Details
L-SerineView Details
56-45-1
