Imiglucerase
- CAS NO.:154248-97-2
- Molecular Weight: 0
- MDL number: MFCD09265408
- Update Date: 2022-12-21 16:56:50
What is Imiglucerase?
Description
Imiglucerase, a mannose-terminated form of human placental glucocerebrosidase produced by recombinant technology, was introduced for the treatment of type-1 Gaucher's disease. lmiglucerase catalyzes the hydrolysis of glucocerebroside and thus prevents accumulation of this lipid in organs and tissues. Compared with the natural enzyme, the modification by removal of the terminal sugars resulted in strong binding of the exposed mannose to lectins expressed on the surface of macrophages, which greatly increases the amount of enzyme taken up by the cells. In 6-month clinical trials, its unequivocal efficacy was shown by improvement in all patients with type-1 Gaucher's disease, leading to an increased hemoglobin levels and platelet counts, decreased incidence of epistaxis and bruising, reduced spleen and liver sizes, and improved skeletal parameters. This enzyme replacement therapy is well tolerated with few mild adverse reactions.
Originator
Genzyme (U.S.A.)
The Uses of Imiglucerase
Enzyme replenisher (glucocerebrosidase).
brand name
Cerezyme (Genzyme).
Clinical Use
Type 1 Gaucher disease is a hereditary condition occurringin about 1:40,000 individuals. It is characterized by a functionaldeficiency in β-glucocerebrosidase enzyme activityand the resulting accumulation of lipid glucocerebroside intissue macrophages, which become engorged and aretermed Gaucher cells. Gaucher cells typically accumulate inthe liver, spleen, and bone marrow and, occasionally, inlung, kidney, and intestine. Secondary hematological sequelaeinclude severe anemia and thrombocytopenia in additionto characteristic progressive hepatosplenomegaly. Skeletalcomplications are common and are frequently the most debilitatingand disabling feature of Gaucher disease. Possibleskeletal complications are osteonecrosis, osteopenia withsecondary pathological fractures, remodeling failure, osteosclerosis,and bone crises.
Cerezyme (Imiglucerase) is a recombinant, macrophagetargetedvariant of human β-glucocerebrosidase, purified fromCHO cells. It catalyzes the hydrolysis of the glycolipid glucocerebrosideto glucose and ceramide following the normaldegradation pathway for membrane lipids.Cerezyme is supplied as a lyophilized powder for reconstitution.The powder should be stored at 2°C to 8°C untilused. The reconstituted product for IV infusion is stable for12 hours at room temperature.
Safety information for Imiglucerase
New Products
Dexamethasone Sodium Phosphate p-Anisaldehyde m-Tolualdehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid Raloxifene EP Impurity D N-Nitroso Nortriptyline Nitroso Irbesartan 2-(p-Tolyl)acetaldehyde Sulfonic acid Impurity 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 5-Chloro-2-Methoxy-4-Picoline 2-Amino-6-nitrobenzoic Acid (6-Nitroanthranilic acid) Cis-3,5 Diacetoxy-1-cyclopentene 3-Bromo-2-Methoxy-4-Methylpyridine 2-Amino-5-Chloro-3-Nitro-6-picoline 3-Methoxy-5-methylpyrazin-2-amine Pentachlorobenzonitrile Budesonide Nicotinic acid RAPAMYCIN Alpha CyclodextrinRelated products of tetrahydrofuran
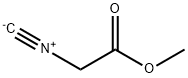
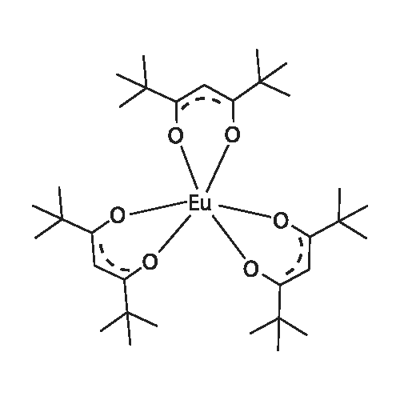

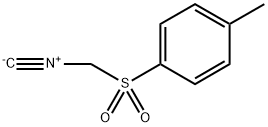
You may like
-
 88660-53-1 >99%View Details
88660-53-1 >99%View Details
88660-53-1 -
 12029-98-0 >98%View Details
12029-98-0 >98%View Details
12029-98-0 -
 1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 -
 Hydrocortisone 50-23-7 >98%View Details
Hydrocortisone 50-23-7 >98%View Details
50-23-7 -
 106-43-4 Parachlorotoluene >99%View Details
106-43-4 Parachlorotoluene >99%View Details
106-43-4 -
 Pyrrolidin >98%View Details
Pyrrolidin >98%View Details
123-75-1 -
 cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
7212-40-0 -
 Para Chloro toluene (PCT) 106-43-4 >99%View Details
Para Chloro toluene (PCT) 106-43-4 >99%View Details
106-43-4
