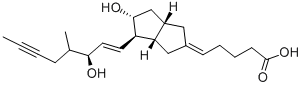
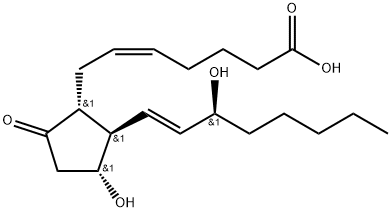
Iloprost
Synonym(s):(E)-(3aS, 4R, 5R, 6aS)-Hexahydro-5-hydroxy-4-[(E)-(3S,4RS)-3-hydroxy-4-methyl-1-octen-6-ynyl]-Δ2(1H),Δ-pentalenevaleric acid;Cilaprost;ZK 36374; (5E)-5-[(3aS,4R,5R,6aS)-Hexahydro-5-hydroxy-4-[(1E,3S)-3-hydroxy-4-methyl-1-octen-6-ynyl]-2(1H)-pentalenylidene]pentanoic acid
- CAS NO.:73873-87-7
- Empirical Formula: C22H32O4
- Molecular Weight: 360.49
- MDL number: MFCD00867145
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-11-08 17:00:29
What is Iloprost?
The Uses of Iloprost
A synthetic analogue of Prostacyclin (PGI2) used to treat pulmonary arterial hypertension (PAH), scleroderma, Raynaud''s phenomenon and ischemia. It acts through elevation of cAMP by binding to the prostacyclin receptor (IP receptor). Iloprost inhibits the ADP, thrombin, and collagen-induced aggregation of human platelets with an ED50 of about 13 nM. In whole animals, iloprost acts as a vasodilator, hypotensive, antidiuretic, and prolongs bleeding time.
The Uses of Iloprost
Treatment of pulmonary hypertension (prostacyclin analogue).
Definition
ChEBI: Iloprost is a carbobicyclic compound that is prostaglandin I2 in which the endocyclic oxygen is replaced by a methylene group and in which the (1E,3S)-3-hydroxyoct-1-en-1-yl side chain is replaced by a (3R)-3-hydroxy-4-methyloct-1-en-6-yn-1-yl group. A synthetic analogue of prostacyclin, it is used as the trometamol salt (generally by intravenous infusion) for the treatment of peripheral vascular disease and pulmonary hypertension. It has a role as a platelet aggregation inhibitor and a vasodilator agent. It is a monocarboxylic acid, a secondary alcohol and a carbobicyclic compound.
brand name
Ventavis (Schering).
General Description
This more chemically and biologically stable derivativeof prostacyclin is available as a solution (10 μg/mL)for nasal inhalation (Ventavis by Actelion) via a preciselycalibrated inhalation device for the treatment of pulmonaryarterial hypertension (PAH). Patients inhale 6 to 8 puffs ofaerosolized iloprost every 2 to 3 hours to produce a directvasodilatory effect on pulmonary blood vessels, therebydecreasing vascular resistance. Side effects of coughing,flushing, headaches, and jaw pain have been most commonlyreported.
Clinical Use
Iloprost is administered as an inhalation solution of a prostacyclin analog for the treatment of NYHA class III and class IV PAH. The drug also can be administered as an IV infusion. It is stable at room temperature and to light, with a body half-life of 30 minutes. Iloprost has approximately 10 times greater potency than prostacyclin as a vasodilator of the pulmonary blood vessels; this greater potency of inhaled iloprost results from coating of the drug on the alveoli of the lungs.
Side Effects
Studies have reported minor side effects, such as coughing, headaches, and jaw pain.
Drug interactions
Potentially hazardous interactions with other drugs
Anticoagulants: enhanced anticoagulant effect and
increased risk of bleeding with heparin, coumarins
and phenindione, as iloprost inhibits platelet
aggregation.
Increased risk of bleeding with NSAIDs, aspirin,
clopidogrel, eptifibatide and tirofiban.
Metabolism
On intravenous infusion iloprost is rapidly cleared from the plasma by oxidation. About 80% of the metabolites are excreted in urine and 20% in the bile.
Properties of Iloprost
| storage temp. | Amber Vial, -20°C Freezer, Under inert atmosphere |
| solubility | Chloroform (Slightly), Methanol (Slightly) |
| form | Solid |
| color | Colourless to Pale Yellow Oil to |
| Stability: | Light Sensitive |
Safety information for Iloprost
Computed Descriptors for Iloprost
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran

You may like
-
 78919-13-8 / 73873-87-7 Iloprost 98%View Details
78919-13-8 / 73873-87-7 Iloprost 98%View Details
78919-13-8 / 73873-87-7 -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
