HALAZEPAM CIV (200 MG) (AS)
- CAS NO.:23092-17-3
- Empirical Formula: C17H12ClF3N2O
- Molecular Weight: 352.74
- EINECS: 245-425-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:15:32
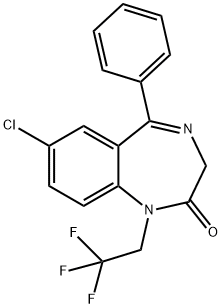
What is HALAZEPAM CIV (200 MG) (AS)?
The Uses of HALAZEPAM CIV (200 MG) (AS)
A benzodiazepine derivative that possesses anxiolytic, anticonvulsant, sedative and skeletal muscle relaxant properties. It is a precursor to Desmethyldiazepam (D291595) and is equal to or more effective than Diazepam (D416855) with a lower frequency of side effects. Controlled Substance.
Background
Halazepam is a benzodiazepine derivative drug exerting anxiolytic, anticonvulsant, sedative, a muscle relaxing effects. It has been shown to be less toxic than chlordiazepoxide or diazepam. This drug is no longer marketed in the United States, and was withdrawn by Schering, its manufacturer, in 2009.
Indications
Used to relieve anxiety, nervousness, and tension associated with anxiety disorders.
Definition
ChEBI: Halazepam is an organic molecular entity.
brand name
Paxipam (Schering).
Metabolism
Hepatic.
Properties of HALAZEPAM CIV (200 MG) (AS)
| Melting point: | 164-166° |
| Boiling point: | 487.8±45.0 °C(Predicted) |
| Density | 1.37±0.1 g/cm3(Predicted) |
| solubility | DMF: 25 mg/ml; DMSO: 25 mg/ml; DMSO:PBS(pH7.2) (1:1): 0.5 mg/ml; Ethanol: 10 mg/ml; Methanol: 1 mg/ml |
| form | A crystalline solid |
| pka | 2.61±0.20(Predicted) |
| EPA Substance Registry System | Halazepam (23092-17-3) |
Safety information for HALAZEPAM CIV (200 MG) (AS)
Computed Descriptors for HALAZEPAM CIV (200 MG) (AS)
New Products
2-(2-aminoethyl) benzenesulfonamide Gemcitabine 3-benzoate impurity Dihydroxyphenylacetone Ornidazole EP impurity D Acetyl amino diol impurity Valsartan Benzyl Ester Impurity B N,N'-Dicyclohexylcarbodiimide 3-Bromo-2-Methyl-5-Nitropyridine 5-Bromo-3-Methyl-2-Pyridinecarboxylic acid 2-Amino-4-Methylpyridine 4-Aminopyridine 4-dimethylaminopyridineRelated products of tetrahydrofuran

You may like
-
 4-(Methylnitrosoamino)-butanal NLT 95%View Details
4-(Methylnitrosoamino)-butanal NLT 95%View Details
339362-86-6 -
 848696-99-1 NLT 95%View Details
848696-99-1 NLT 95%View Details
848696-99-1 -
 Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
1384752-15-1 -
 615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 -
 175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 -
 S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
1346605-15-9 -
 Valsartan Related Compound A NLT 95%View Details
Valsartan Related Compound A NLT 95%View Details
137862-87-4 -
 Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
124750-49-8
