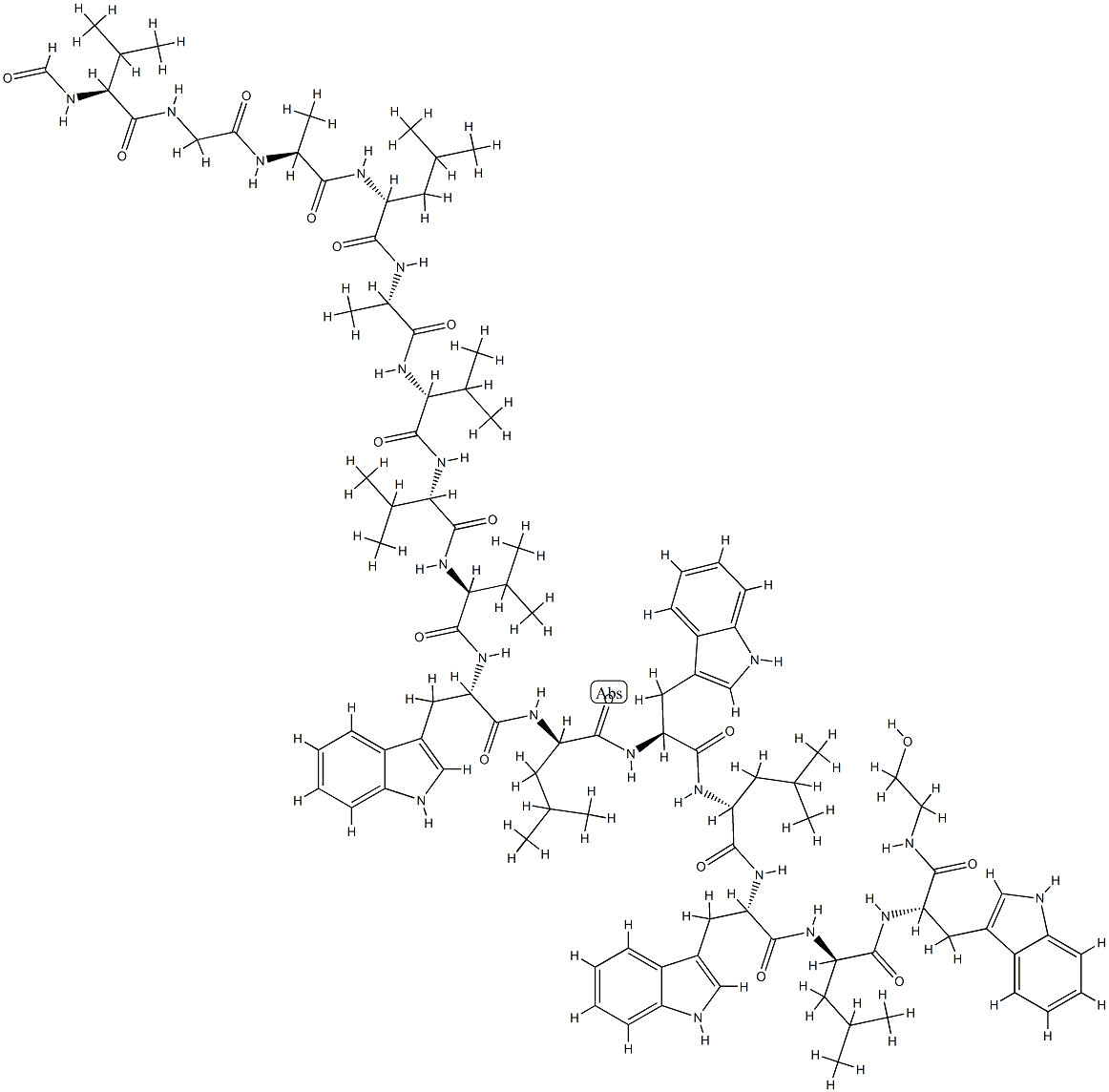
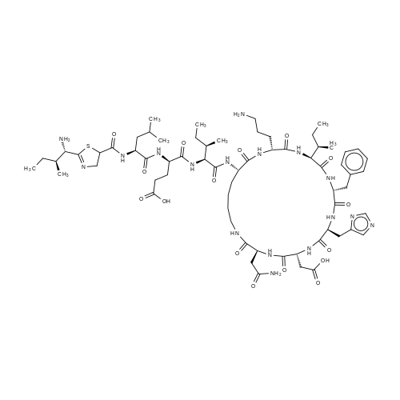
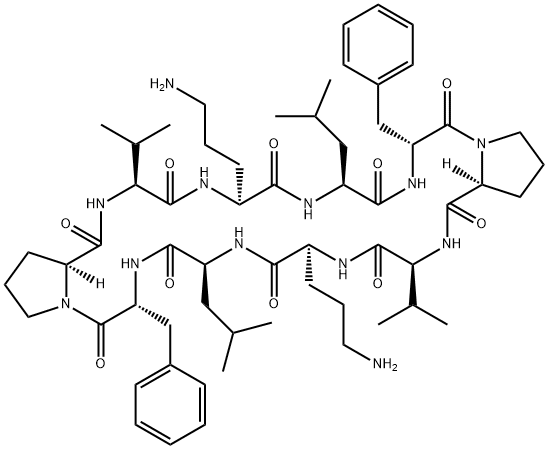
GRAMICIDIN C
- CAS NO.:113-73-5
- Empirical Formula: C60H92N12O10
- Molecular Weight: 1141.45
- MDL number: MFCD00466945
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-11-05 16:33:38
What is GRAMICIDIN C?
Description
Gramicidin S was found in the culture broth of Bacillus brevis by Gause of the USSR Academy of Medical Sciences in 1944. It was later found by Otani of Osaka University in the cells of the Nagano strain of the same species. The antibiotic is a basic peptide showing strong activity against gram-positive bacteria and considerable activity against gramnegative cocci and Mycobacterium. Gramicidin S is rather toxic but is not absorbed orally; it is used topically as an ointment or as eye or ear drops in combination with other antibacterial drugs.
Originator
Gramoderm,Schering,US,1949
Definition
A cyclic peptide that acts on gram-positive bacteria.
Manufacturing Process
5 lb of acid precipitated solid (Hotchkiss, Advances in Enzymology, pages 157-
158) from 30 gal of tyrothricin fermentation liquor containing about 40 g
(2%)of tyrothricin were extracted with 12 liters of absolute ethyl alcohol and
filtered. The filtrate was evaporated in vacuo to 1 liter, and the concentrate
extracted twice with 1 liter of pentane. The pentane layers were discarded.40 g of decolorizing charcoal were added to the pentane-extracted filtrate and
filtered off.
To 500 ml of the charcoal-treated filtrate were added 200 ml benzene and 300
ml water, the whole shaken thoroughly, centrifuged, and the benzene layer
separated. This treatment of the charcoal-treated filtrate was repeated twice,
all benzene fractions were combined and evaporated in vacuo.
200 ml of absolute acetone were added to the residue and concentrated by
boiling to 150 ml. The concentrate was refrigerated overnight. The crystals
which had formed in the concentrate were filtered off, and the mother liquor
concentrated first to 50 ml and then to 25 ml, the two concentrates
refrigerated overnight, and the formed crystals filtered off. Total yield of
crystalline gramicidin was 3.85 g = 19.2% of estimated tyrothricin in the
initial material.
The combined crystal crops were redissolved in 50 ml absolute acetone, and
the solution refrigerated overnight. After filtering, the formed crystals were
dried in vacuo. The total yield of crystalline gramicidin thus obtained was 2.5
g.
Therapeutic Function
Antibacterial
Purification Methods
Gramicidin S crystallises from EtOH. The di-HCl crystallises from EtOH (+ few drops of HCl) with m 277-278o (see below). [NMR: Gibbons et al. Nature 227 840 1970, Beilstein 26 III/IV 4273.]
Properties of GRAMICIDIN C
| Melting point: | 265-270 °C (decomp)(Solv: ethanol (64-17-5); ethyl ether (60-29-7)) |
| Boiling point: | 827.42°C (rough estimate) |
| Density | 1.0265 (rough estimate) |
| refractive index | 1.6000 (estimate) |
| pka | 13.00±0.70(Predicted) |
| form | Solid |
| color | White to off-white |
| Water Solubility | 0.14g/L(28 ºC) |
Safety information for GRAMICIDIN C
Computed Descriptors for GRAMICIDIN C
| InChIKey | IUAYMJGZBVDSGL-XNNAEKOYSA-N |
New Products
2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid 8-Bromo-3-methyl xanthine Maleic hydrazide o-Anisaldehyde 4-Bromobenzaldehyde 3-Fluorobenzaldehyde 2-Chlorobenzaldehyde 3-Amino-3-(3-fluorophenyl)propanoic acid 1-Propyl-4-piperidone 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2-Fluoro-6-iodobenzoic acid 3-Pyridineacrylic acid 1-Boc-4-cyanopiperidine ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate Vardenafil Bis-sulphonamide(Dimer) Nitroso Irbesartan N-Nitroso Nortriptyline Sulfonic acid Impurity 2-(p-Tolyl)acetaldehyde 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran
You may like
-
 Deuterated Styrene-d8 19361-62-7 97-99%View Details
Deuterated Styrene-d8 19361-62-7 97-99%View Details
19361-62-7 -
 1187989-89-4 97-99%View Details
1187989-89-4 97-99%View Details
1187989-89-4 -
 (S)-2-Bromobutanoic acid 97-99%View Details
(S)-2-Bromobutanoic acid 97-99%View Details
32659-49-7 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 methyl 3-amino-4-formylbenzoate 97-99%View Details
methyl 3-amino-4-formylbenzoate 97-99%View Details
212322-17-3 -
![3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%View Details
3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%View Details
1620885-59-7 -
![(13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) (13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%View Details
(13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%View Details
2590-41-2 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2