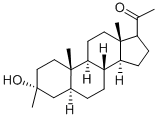
GANAXOLONE
- CAS NO.:38398-32-2
- Empirical Formula: C22H36O2
- Molecular Weight: 332.52
- MDL number: MFCD09971088
- EINECS: 601-766-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-01-30 17:41:12
What is GANAXOLONE?
Absorption
Following oral administration, maximum plasma concentrations are reached within 2 to 3 hours.
Toxicity
Experience with ganaxolone overdose is limited to a single report of an unintentional overdose in a pediatric patient, in which the patient received ten-fold the prescribed dose. The patient was hospitalized for evaluation and recovered. No specific information is available regarding treatment of ganaxolone overdose - patients suspected of overdosage should be closely monitored and receive standard supportive care.
Description
Ganaxolone is a 3β-methylated synthetic analog of the neurosteroid allopregnanolone that allosterically enhances GABAA receptor current. At nanomolar concentrations, it can potentiate GABA-evoked currents at GABAA receptor complexes containing α1, α2, α3, β2, and γ2L subunits expressed in Xenopus oocytes. Ganaxolone has been shown to have a broad range of antiseizure activity in animal epilepsy models and human clinical trials.
The Uses of GANAXOLONE
Ganaxolone, is synthetic neuroactive steroid, which possess sedative, anxiolytic, and anticonvulsant properties. It is also a potent and selective positive allosteric modulator of GABAA receptors. It is also shown to be used for the treatment of epilepsy.
The Uses of GANAXOLONE
Treatment of epilepsy and migraine.
Background
Ganaxolone is the 3β-methylated synthetic analog of allopregnanolone, a metabolite of progesterone. Ganaxolone belongs to a class of compounds referred to as neurosteroids. Endogenous neurosteroids, which comprise certain metabolites of progesterone and deoxycorticosterone, bind potently and specifically to GABAA receptors to enhance their inhibitory effects, and are thus known to have anxiolytic, analgesic, anticonvulsant, sedative, hypnotic, and anesthetic properties.
Ganaxolone, similar to its endogenous counterparts, is a positive allosteric modulator of GABAA receptors. It was approved under the brand name ZTALMY by the US FDA in March 2022 for the treatment of seizures associated with CDKL5 deficiency disorder (CDD), becoming the first FDA-approved treatment indicated specifically for CDD. In July 2023, ganaxolone was also approved under the same brand name and for the same indication by the EMA.
What are the applications of Application
Ganaxolone is a potent stereoselective modulator of GABAA receptors
Indications
Ganaxolone is indicated for the treatment of seizures associated with cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder (CDD) in patients ≥2 years old by the FDA. It is also approved as an adjuvant treatment for the same condition in patients aged 2 to 17, although it may be continued in patients 18 years old or older, by the EMA.
Definition
ChEBI: Ganaxolone is a corticosteroid hormone.
Biological Activity
Potent positive allosteric modulator of GABA A receptors. Enhances GABA-evoked chloride currents in Xenopus oocytes expressing GABA A receptors (EC 50 values are 94, 122 and 213 nM for α 2 β 1 γ 2 L , α 3 β 1 γ 2 L and α 1 β 1 γ 2 L receptors respectively). Exerts anticonvulsive effects in a broad range of animal seizure models.
Biochem/physiol Actions
Ganaxolone (3alpha-hydroxy-3beta-methyl-5alpha-pregnane-20-one) is a positive allosteric modulator of the GABAA receptor subtype; synthetic analog of the endogenous neurosteroid allopregnanolone; effective against chemically induced seizures in rats and mice. Ganaxolone is an orally active analog of allopregnanolone that is not converted to the hormonally active 3-keto form. The enhanced anticonvulsant potency of ganaxolone after neurosteroid withdrawal supports the use of ganaxolone as a specific treatment for perimenstrual catamenial epilepsy.
Pharmacokinetics
Ganaxolone - like other neurosteroids - lacks classical hormonal activity, and instead appears to exert its effects directly via modulation of GABAA receptors. Similar to other antiepileptic drugs, ganaxolone has been associated with significant somnolence and sedation - patients should be instructed to use caution when operating heavy machinery (e.g. driving). In addition, antiepileptic drugs may increase the risk of suicidal behaviour and ideation, although this risk has not been documented directly in patients taking ganaxolone. When considering ganaxolone therapy, clinicians should balance the risk of suicidal thoughts or behaviours with the risk of untreated illness.
Ganaxolone drug scheduling is currently under review by the US Drug Enforcement Administration (DEA), but it appears to carry some potential for abuse. Clinicians should consider assessing for a history of drug abuse when deciding to initiate therapy with ganaxolone.
Metabolism
Ganaxolone is extensively metabolized, primarily by CYP3A4/5 and, to a lesser extent, CYP2B6, CYP2C19, and CYP2D6. While data regarding ganaxolone metabolism are lacking, a 16-OH metabolite generated via CYP3A4 metabolism has been identified as one of its major metabolites.
Storage
+4°C
Properties of GANAXOLONE
| Melting point: | 190-192° |
| Boiling point: | 434.8±18.0 °C(Predicted) |
| alpha | D +103° |
| Density | 1.036±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | DMSO: ~4.2 mg/mL |
| form | solid |
| pka | 15.18±0.70(Predicted) |
| color | white |
Safety information for GANAXOLONE
Computed Descriptors for GANAXOLONE
New Products
Dexamethasone Sodium Phosphate p-Anisaldehyde m-Tolualdehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid Raloxifene EP Impurity D N-Nitroso Nortriptyline Nitroso Irbesartan 2-(p-Tolyl)acetaldehyde Sulfonic acid Impurity 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 5-Chloro-2-Methoxy-4-Picoline 2-Amino-6-nitrobenzoic Acid (6-Nitroanthranilic acid) Cis-3,5 Diacetoxy-1-cyclopentene 3-Bromo-2-Methoxy-4-Methylpyridine 2-Amino-5-Chloro-3-Nitro-6-picoline 3-Methoxy-5-methylpyrazin-2-amine Pentachlorobenzonitrile Budesonide Nicotinic acid RAPAMYCIN Alpha CyclodextrinRelated products of tetrahydrofuran
You may like
-
 Ganaxolone CAS 38398-32-2View Details
Ganaxolone CAS 38398-32-2View Details
38398-32-2 -
 12029-98-0 >98%View Details
12029-98-0 >98%View Details
12029-98-0 -
 1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 -
 Hydrocortisone 50-23-7 >98%View Details
Hydrocortisone 50-23-7 >98%View Details
50-23-7 -
 106-43-4 Parachlorotoluene >99%View Details
106-43-4 Parachlorotoluene >99%View Details
106-43-4 -
 Pyrrolidin >98%View Details
Pyrrolidin >98%View Details
123-75-1 -
 cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
7212-40-0 -
 Para Chloro toluene (PCT) 106-43-4 >99%View Details
Para Chloro toluene (PCT) 106-43-4 >99%View Details
106-43-4