FERULENOL
- CAS NO.:6805-34-1
- Empirical Formula: C24H30O3
- Molecular Weight: 366.49
- MDL number: MFCD02183471
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 14:59:19
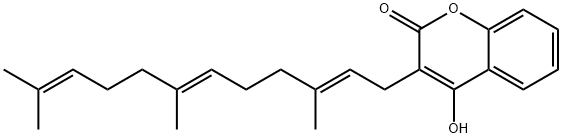
What is FERULENOL?
The Uses of FERULENOL
Ferulenol is antibiotic useful against mycobacteria similar to taxol.
What are the applications of Application
Ferulenol is antibiotic useful against mycobacteria similar to taxol
Biological Activity
ferulenol is a prenylated 4-hydroxycoumarin derivative from ferula communis var. genuine with haemorrhagic action [1]. it has been demonstrated that ferulenol exihibits potent antimycobacterial activity [2].
in vitro
ferulenol stimulated tubulin polymerization in the absence of gtp, with a less extensive polymerization profile at 100 pm concentration. ferulenol decreased radiolabeled colchicine bound by tubulin in a dose-dependent manner. ferulenol altered the normal nuclear morphology of mcf-7 cells. treatment with ferulenol (100 nm and 1 μm) for 24h induced a dose-dependent reduction of cell viability [3]
in vivo
in albino mice, the acute ld50s of ferulenol by single po or ip were 2,100 and 319 mg/kg bw, respectively. three days after ferulenol administration, dosed animals showed hypoprothrombinemia with internal and external hemorrhages. male mice were more sensitive to intoxication than females [4].
Storage
+4°C
References
[1] lamnaouer d, bodo b, martin m t, et al. ferulenol and ω-hydroxyferulenol, toxic coumarins from ferula communis var. genuina[j]. phytochemistry, 1987, 26(6): 1613-1615.
[2] e. mamoci, i. cavoski, v. simone, et al. chemical composition and in vitro activity of plant extracts from ferula communis and dittrichia viscosa against postharvest fungi. molecules 16(3), 2609-2625 (2011).
[3] bocca c, gabriel l, bozzo f, et al. microtubule-interacting activity and cytotoxicity of the prenylated coumarin ferulenol[j]. planta medica, 2002, 68(12): 1135-1137.
[4] fraigui o, lamnaouer d, faouzi m y. acute toxicity of ferulenol, a 4-hydroxycoumarin isolated from ferula communis l[j]. veterinary and human toxicology, 2002, 44(1): 5-7.
Properties of FERULENOL
| Melting point: | 55-56℃ |
| Boiling point: | 525.3±50.0 °C(Predicted) |
| Density | 1.070±0.06 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | Soluble in DMSO |
| pka | 4.50±1.00(Predicted) |
| form | Off-white solid. |
| color | White to off-white |
Safety information for FERULENOL
Computed Descriptors for FERULENOL
New Products
2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid 8-Bromo-3-methyl xanthine Maleic hydrazide o-Anisaldehyde 4-Bromobenzaldehyde 3-Fluorobenzaldehyde 2-Chlorobenzaldehyde 3-Amino-3-(3-fluorophenyl)propanoic acid 1-Propyl-4-piperidone 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2-Fluoro-6-iodobenzoic acid 3-Pyridineacrylic acid 1-Boc-4-cyanopiperidine ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate Vardenafil Bis-sulphonamide(Dimer) Nitroso Irbesartan N-Nitroso Nortriptyline Sulfonic acid Impurity 2-(p-Tolyl)acetaldehyde 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran
You may like
-
 Deuterated Styrene-d8 19361-62-7 97-99%View Details
Deuterated Styrene-d8 19361-62-7 97-99%View Details
19361-62-7 -
 1187989-89-4 97-99%View Details
1187989-89-4 97-99%View Details
1187989-89-4 -
 (S)-2-Bromobutanoic acid 97-99%View Details
(S)-2-Bromobutanoic acid 97-99%View Details
32659-49-7 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 methyl 3-amino-4-formylbenzoate 97-99%View Details
methyl 3-amino-4-formylbenzoate 97-99%View Details
212322-17-3 -
![3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%View Details
3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%View Details
1620885-59-7 -
![(13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) (13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%View Details
(13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%View Details
2590-41-2 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2


