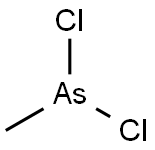
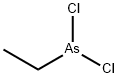
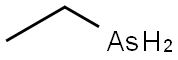Ethyldichloroarsine
- CAS NO.:598-14-1
- Empirical Formula: C2H5AsCl2
- Molecular Weight: 174.89
- EINECS: 209-919-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-01 16:59:24
What is Ethyldichloroarsine?
Chemical properties
Colorless, mobile liquid; becomes yellowish under the action of light and air. Fruitlike odor (high dilution). Decomposed by water. Attacks brass but not iron (dry). Soluble in alcohol, benzene, ether, and water.
Chemical properties
A colorless mobile liquid. Acrid, biting, irritating, fruit-like odor
The Uses of Ethyldichloroarsine
Military poison.
General Description
A colorless liquid with a fruity biting irritating odor. Vapors irritate eyes and skin. Very toxic by inhalation. Boiling point 168°F. Denser than water and vapors are heavier than air. Vapor pressure 2.3 mm at 71°F.
Air & Water Reactions
Reacts with water or moist air to form hydrochloric acid or fumes of hydrogen chloride.
Reactivity Profile
ETHYL DICHLOROARSINE is a reducing agent. Reacts rapidly and dangerously with oxygen and with other oxidizing agents, even weak ones. Likely to ignite on contact with alcohols. Incompatible with acids, alcohols, amines, and aldehydes. May be ignited by heat, sparks or flames. May re-ignite after fire is extinguished.
Hazard
Toxic by ingestion, inhalation, and skin absorption; strong irritant.
Health Hazard
Highly toxic, may be fatal if inhaled, swallowed or absorbed through skin. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Containers may explode when heated. Runoff may pollute waterways.
Safety Profile
A human poison by inhalation. Experimentally, a deadly poison by inhalation and subcutaneous routes, and probably by ingestion. A severe irritant. A mdltary poison gas. Can react with oxidzing materials. Will react with water or steam to produce toxic and corrosive fumes. Dangerous; on contact with acid or acid fumes it emits highly toxic fumes of Cl-, As, and phosgene. See also ARSENIC COMPOUNDS.
Potential Exposure
Organoarsenic compound, Organometallic.
Shipping
UN1892 Ethyldichloroarsine Hazard class: 6.1; Labels: 6.1-Poison Inhalation Hazard; Inhalation Hazard Zone B. Military driver shall be given full and complete information regarding shipment and conditions in case of emergency. AR 50-6 deals specifically with the shipment of chemical agents. Shipments of agent will be escorted in accordance with AR 740-32
Incompatibilities
Reacts with water or moist air releasing hydrochloric acid and/or corrosive fumes of hydrogen chloride (HCl). A strong reducing agent; incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, and epoxides. Attacks some metals; brass, but not ferrous metals
Waste Disposal
Use a licensed professional waste disposal service to dispose of this material. Dissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal, state, and local environmental regulations must be observed.
Properties of Ethyldichloroarsine
| Melting point: | -64.9°C |
| Boiling point: | 155.37°C (estimate) |
| Density | 1.7010 |
| refractive index | 1.5550 |
| form | liquid |
| CAS DataBase Reference | 598-14-1 |
| EPA Substance Registry System | Ethyldichloroarsine (598-14-1) |
Safety information for Ethyldichloroarsine
Computed Descriptors for Ethyldichloroarsine
New Products
Dexamethasone Sodium Phosphate 2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid 8-Bromo-3-methyl xanthine Maleic hydrazide m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 2-Fluoro-6-iodobenzoic acid 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 1-Boc-4-cyanopiperidine 3-Pyridineacrylic acid Abiraterone Ethyl Ether Vardenafil Bis-sulphonamide(Dimer) 1-nitro-3,5-dimethyl adamantine (NMEM) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5Related products of tetrahydrofuran
You may like
-
 141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 -
 Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
27143-07-3 -
 94-71-3 98%View Details
94-71-3 98%View Details
94-71-3 -
 Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
33045-52-2 -
 88965-00-8 98%View Details
88965-00-8 98%View Details
88965-00-8 -
 3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
107228-51-3 -
 4640-67-9 98%View Details
4640-67-9 98%View Details
4640-67-9 -
![2-[(2-Ethoxyphenoxy)methyl]oxirane 98%](https://img.chemicalbook.in//Content/image/CP5.jpg) 2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
5296-35-5