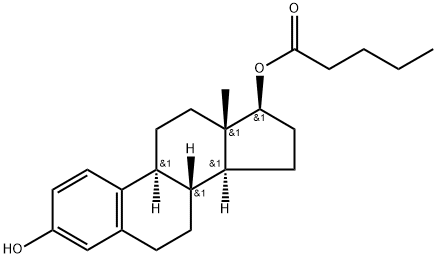
estra-1,3,5(10)-triene-3,17beta-diol diheptanoate
- CAS NO.:7732-97-0
- Empirical Formula: C32H48O4
- Molecular Weight: 496.72112
- MDL number: MFCD03695580
- EINECS: 231-792-8
- SAFETY DATA SHEET (SDS)
- Update Date: 2022-12-21 16:56:50
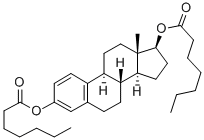
What is estra-1,3,5(10)-triene-3,17beta-diol diheptanoate?
The Uses of estra-1,3,5(10)-triene-3,17beta-diol diheptanoate
Estradiol Dienanthate is a metabolite of estradiol (E888000), which is the major estrogen secreted by the premenopausal ovary.
Indications
Estradiol dienanthate is not currently available in any FDA or Health Canada approved products.
Background
Estradiol Dienanthate is a pro-drug ester of Estradiol, a naturally occurring hormone that circulates endogenously within the human body. Estradiol is the most potent form of all mammalian estrogenic steroids and acts as the major female sex hormone. As a pro-drug of estradiol, estradiol benzoate therefore has the same downstream effects within the body through binding to the Estrogen Receptor (ER) including ERα and ERβ subtypes, which are located in various tissues including in the breasts, uterus, ovaries, skin, prostate, bone, fat, and brain.
Estradiol is commonly produced with an ester side-chain as endogenous estradiol has very low oral bioavailability on its own (2-10%). First-pass metabolism by the gut and the liver quickly degrades the estradiol molecule before it gets a chance to enter systemic circulation and exert its estrogenic effects . Esterification of estradiol aims to improves absorption and bioavailability after oral administration (such as with Estradiol valerate) or to sustain release from depot intramuscular injections (such as with Estradiol Cypionate) through improved lipophilicity. Following absorption, the esters are cleaved, resulting in the release of endogenous estradiol, or 17β-estradiol. Ester pro-drugs of estradiol are therefore considered to be bioidentical forms of estrogen .
Estradiol dienanthate is not currently available in any commercially available products in Canada or the US.
Pharmacokinetics
Estradiol, the principal intracellular human estrogen, is substantially more active than its metabolites, estrone and estriol, at the cellular level.
Metabolism
Exogenous estrogens are metabolized using the same mechanism as endogenous estrogens. Estrogens are partially metabolized by cytochrome P450.
Safety information for estra-1,3,5(10)-triene-3,17beta-diol diheptanoate
New Products
Dexamethasone Sodium Phosphate p-Anisaldehyde m-Tolualdehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid Raloxifene EP Impurity D N-Nitroso Nortriptyline Nitroso Irbesartan 2-(p-Tolyl)acetaldehyde Sulfonic acid Impurity 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 5-Chloro-2-Methoxy-4-Picoline 2-Amino-6-nitrobenzoic Acid (6-Nitroanthranilic acid) Cis-3,5 Diacetoxy-1-cyclopentene 3-Bromo-2-Methoxy-4-Methylpyridine 2-Amino-5-Chloro-3-Nitro-6-picoline 3-Methoxy-5-methylpyrazin-2-amine Pentachlorobenzonitrile Budesonide Nicotinic acid RAPAMYCIN Alpha CyclodextrinRelated products of tetrahydrofuran




You may like
-
 88660-53-1 >99%View Details
88660-53-1 >99%View Details
88660-53-1 -
 12029-98-0 >98%View Details
12029-98-0 >98%View Details
12029-98-0 -
 1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 -
 Hydrocortisone 50-23-7 >98%View Details
Hydrocortisone 50-23-7 >98%View Details
50-23-7 -
 106-43-4 Parachlorotoluene >99%View Details
106-43-4 Parachlorotoluene >99%View Details
106-43-4 -
 Pyrrolidin >98%View Details
Pyrrolidin >98%View Details
123-75-1 -
 cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
7212-40-0 -
 Para Chloro toluene (PCT) 106-43-4 >99%View Details
Para Chloro toluene (PCT) 106-43-4 >99%View Details
106-43-4
