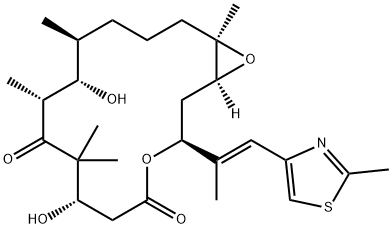
Epothilone A
Synonym(s):EpoA
- CAS NO.:152044-53-6
- Empirical Formula: C26H39NO6S
- Molecular Weight: 493.66
- MDL number: MFCD26960884
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-22 18:57:36
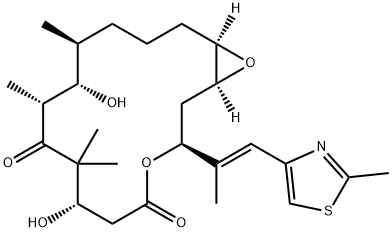
What is Epothilone A?
Description
Epothilone-A is the first of a new class of compounds that can fight cancer cells that resist conventional chemotherapy. Altough the epothilones work in a manner similar to paclitaxel, they have simpler structures, are easier to make, and dissolve better in water.
The Uses of Epothilone A
The epothilones are a novel class of antineoplastic agents possessing antitubulin activity. Epothilone A, B isolated from Sorangium cellulosum was associated with cell cycle arrest at G2/M transition and apoptosis that was resisted with overexpression of β-tubulin and P-glycoprotein in human bladder carcinoma cell.
The Uses of Epothilone A
Epothilone A is a microtubule inhibitor isolated from the myxobacteria, Sorangium cellulosum. Epothilone A acts by stabilising microtubule formation at the taxol binding site, and causes cell cycle arrest at the G2/M transition, leading to cytotoxicity. Epothilone A has been investigated in clinical trials as an antitumour agent.
What are the applications of Application
Epothilone A is an antitubulin agent
Definition
ChEBI: An epithilone that is epothilone C in which the double bond in the macrocyclic lactone ring has been oxidised to the corresponding epoxide (the 13R,14S diastereoisomer).
Biological Activity
epothilone a (epo a) is a naturally occurring microtubule-stabilizing macrolide that was first isolated from the myxobacterium sorangium cellulosum. the ic50 value of epothilone a in hct-116 cell line is 4.4 nm [1].it has been found that the skov-3 human ovarian cancer cells were susceptible to epothilone a with ic50 value of 20.4 ± 1.4 nm [2]. the antiproliferative capacity of epothilone a in skov-3 cell line (measured as ic50 after 72 h continuous treatment) was six times greater than that of ptx’s. besides, epothilone a induced time coursedependent apoptosis and necrosis [2].
References
[1] regueiro-ren a1, borzilleri rm, zheng x, kim sh, johnson ja, fairchild cr, lee fy, long bh, vite gd. synthesis and biological activity of novel epothilone aziridines. org lett. 2001 aug 23;3(17):2693-6.
[2] rogalska a1, marczak a, gajek a, szwed m, śliwińska a, drzewoski j, jóźwiak z. induction of apoptosis in human ovarian cancer cells by new anticancer compounds, epothilone a and b. toxicol in vitro. 2013 feb;27(1):239-49.
Properties of Epothilone A
| Melting point: | 95℃ |
| Boiling point: | 683.3±55.0 °C(Predicted) |
| alpha | D21 -47.1° (c = 1.0 in methanol) |
| Density | 1.143 |
| storage temp. | -20°C |
| solubility | Chloroform (Slightly), Ethyl Acetate (Slightly) |
| form | solid |
| pka | 13.57±0.70(Predicted) |
| color | White to Off-White |
| Stability: | Stable under recommended storage conditions., Stable Under Recommended Storage C |
Safety information for Epothilone A
Computed Descriptors for Epothilone A
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloropyridazine 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 6-Methyl-2-pyridinemethanol 2-Pyridinecarboxaldehdye tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 1-Indanone 2-Chloromethyl-6-methyl-pyridine 4-Morpholinoaniline Abiraterone Ethyl Ether 1-nitro-3,5-dimethyl adamantine (NMEM) Vardenafil Bis-sulphonamide(Dimer) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran
![(1S,3S,7S,10R,11S,12S,16R)-3-[(1E)-2-[2-(Aminomethyl)-4-thiazolyl]-1-methylethenyl]-7,11-dihydroxy-8,8,10,12,16-pentamethyl-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione](https://img.chemicalbook.in/CAS/GIF/280578-49-6.gif)

You may like
-
 (−)-Epothilone A CAS 152044-53-6View Details
(−)-Epothilone A CAS 152044-53-6View Details
152044-53-6 -
![3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
2304754-51-4 -
 1126-74-5 99%+View Details
1126-74-5 99%+View Details
1126-74-5 -
 2-Fluoro-6-iodobenzoic acid 99%+View Details
2-Fluoro-6-iodobenzoic acid 99%+View Details
111771-08-5 -
 120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 -
 4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
138163-08-3 -
![ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
313544-31-9 -
 1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
91419-52-2
