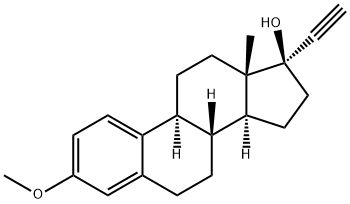
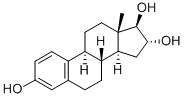
EPIMESTROL
- CAS NO.:7004-98-0
- Empirical Formula: C19H26O3
- Molecular Weight: 302.41
- MDL number: MFCD00198991
- EINECS: 230-278-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-04 17:34:41
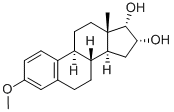
What is EPIMESTROL?
Originator
Stimovul,Organon,W. Germany,1976
The Uses of EPIMESTROL
Anterior pituitary activator.
Definition
ChEBI: Epimestrol is a steroid. It derives from a hydride of an estrane.
Manufacturing Process
Reduction of 16-keto-17(α)-hydroxyestratrienol-3-methyl to 16,17- dihydroxyestratrienol-3-methyl ether: A solution of 800 mg of the alpha ketol methyl ether in 100 cc of ethanol and 10 cc of acetic acid was carefully maintained at 40°C (water bath), and 200 g of freshly prepared sodium amalgam (2%) were added in small pieces with efficient swirling. Before all ofthe amalgam had been added, a precipitation of sodium acetate occurred, and at this point an additional 100 cc of 50% acetic acid were added. After all the reducing agent had been added, the mixture was transferred to a separatory funnel with ether and water. The mercury plus aqueous phase was separated, after partitioning, from the ether; the latter may be further washed with water, with 0.5N sodium hydroxide, and again with water to purify the alpha glycol. Evaporation of the ethereal phase yielded a crystalline residue of the isomeric transoid (16(β),17(α)-dihydroxy-steroid-3-methyl ether and cisoid 16(α),17(α)dihydroxy-steroid-3-methyl ether.
Therapeutic Function
Anterior pituitary activator
Properties of EPIMESTROL
| Melting point: | 158-160° |
| alpha | D20 +48° (chloroform) |
| Boiling point: | 450.8±45.0 °C(Predicted) |
| Density | 1.185±0.06 g/cm3(Predicted) |
| pka | 14.53±0.60(Predicted) |
Safety information for EPIMESTROL
Computed Descriptors for EPIMESTROL
New Products
Hendecanoic acid 2-Amino-5-cyanopyridine 2-Bromo-5-cyanopyridine DL-3-Amino-3-(2-methoxyphenyl)propionic acid 3-Hydroxypropionitrile 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4,4-Diethoxybutanenitrile 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside 6-(4-Amino-2,6-dichlorophenoxy)-4-isopropylpyridazin-3(2H)-one Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 2-Bromo-6-fluoroaniline 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 2-Aminobenzo[d]thiazol-4-ol Methyl 2-aminobutanoate hydrochloride 3-Fluoro-6-methylpicolinonitrile 1-(4-Aminopiperidin-1-yl)-3,3,3-trifluoropropan-1-one 5-chloro-2-(4-methoxybenzyl)-4-(trifluoromethyl)pyridazin-3(2H)-one 3-cyanocyclohexane-1-carboxylic acidRelated products of tetrahydrofuran


You may like
-
 103095-63-2 DL-3-Amino-3-(2-methoxyphenyl)propionic acid 98+View Details
103095-63-2 DL-3-Amino-3-(2-methoxyphenyl)propionic acid 98+View Details
103095-63-2 -
 3-Hydroxypropionitrile 98+View Details
3-Hydroxypropionitrile 98+View Details
109-78-4 -
 103095-63-2 DL-3-Amino-3-(2-methoxyphenyl)propionic acid 98+View Details
103095-63-2 DL-3-Amino-3-(2-methoxyphenyl)propionic acid 98+View Details
103095-63-2 -
 117391-48-7 98+View Details
117391-48-7 98+View Details
117391-48-7 -
 3-Hydroxypropionitrile 98+View Details
3-Hydroxypropionitrile 98+View Details
109-78-4 -
 103095-63-2 DL-3-Amino-3-(2-methoxyphenyl)propionic acid 98+View Details
103095-63-2 DL-3-Amino-3-(2-methoxyphenyl)propionic acid 98+View Details
103095-63-2 -
 117391-48-7 98+View Details
117391-48-7 98+View Details
117391-48-7 -
 3-Hydroxypropionitrile 98+View Details
3-Hydroxypropionitrile 98+View Details
109-78-4
