ENNIATIN FROM MICROBIAL SOURCE
- CAS NO.:11113-62-5
- Empirical Formula: C36H63N3O9.C35H61N3O9.C34H59N3O9.C33H57N3O9
- Molecular Weight: 2643.46
- MDL number: MFCD00214209
- Update Date: 2026-04-21 10:31:43
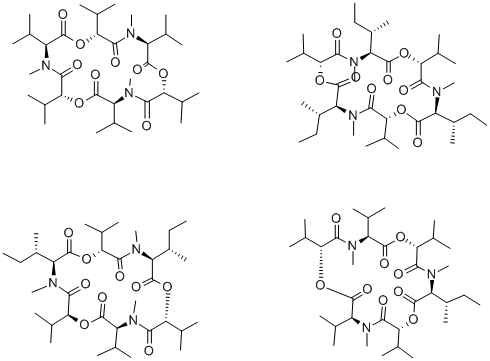
What is ENNIATIN FROM MICROBIAL SOURCE?
The Uses of ENNIATIN FROM MICROBIAL SOURCE
Enniatins are a complex of depsipeptides produced by several Fusarium species. Typically, the complex contains 4 major components: A, A1, B and B1 together with minor amounts of enniatins C, D, E and F. The enniatins act as ionophores. Recently their effects on acyl-CoA cholesterol transferase, as nematocides and the selectivity of their antitumour action have received more focus.
The Uses of ENNIATIN FROM MICROBIAL SOURCE
Enniatin complex is a complex mixture of Fusarium produced depsipeptide ionophores.This compound can be used to control contamination in fermentation of gaseous substrates.
What are the applications of Application
Enniatin complex is a complex mixture of Fusarium produced depsipeptide ionophores
Biological Activity
enniatins, analogues of beauvericin, is a regular cyclic hexadepsipeptide isolated from fusarium species of fungis, also they have been isolated from other genera, such as verticillium and halosarpheia [1]. they have been involved in many biological activities, such as antiinsectan, antifungal, antibiotic and cytotoxic [1].
in vitro
fusafungine is a mixture of several enniatins with bacteriostatic activity against most micro-organisms responsible for infections and superinfections of the respiratory tract. fusafungine has been developed for treatment of upper respiratory tract infections by oral and/or nasal inhalation. fusafungine in low concentrations down-regulated the expression of intercellular adhesion molecule-1 (icam-1) by activated macrophages, inhibited the production of the proinflammatory cytokines il-1, tnfα and il-6 and inhibited the release of oxygen free radicals by inflammatory macrophages without altering their phagocytic activity. fusafungine also inhibited t-cell activation and proliferation, and the synthesis of ifn-γ by activated t cells [2]. exposure 8 h to enniatins at nanomolar concentrations significantly stimulated cell proliferation. at low micromolar concentrations, enniatins exihibited profound apoptosis-inducing effects against various human cancer cell types. in the fluorescence-activated cell sorting analysis, enniatins induced cell cycle arrest in the g0/g1 phase. in human cancer cells, elevated enn concentrations induced profound p53-dependent cytostatic and p53-independent cytotoxic activities [3]. enniatin easily incorporated into the cell membrane in which it formed cation-selective pores [4].
Purification Methods
It is a cyclic peptidic ester antibiotic which is recrystallised from EtOH/water but is deactivated in alkaline solution. [Ovchinnikov & Ivanov in The Proteins (Neurath and Hill eds) Academic Press, NY, Vol V pp. 365 and 516 1982, Tomeda et al. J Antibiot 45 1626 1992.]
References
[1] sy-cordero a a, pearce c j, oberlies n h. revisiting the enniatins: a review of their isolation, biosynthesis, structure determination and biological activities[j]. the journal of antibiotics, 2012, 65(11): 541-549.
[2] german-fattal m. fusafungine, an antimicrobial with anti-inflammatory properties in respiratory tract infections[j]. clinical drug investigation, 2001, 21(9): 653-670.
[3] dornetshuber r, heffeter p, kamyar m r, et al. enniatin exerts p53-dependent cytostatic and p53-independent cytotoxic activities against human cancer cells[j]. chemical research in toxicology, 2007, 20(3): 465-473.
[4] kamyar m, rawnduzi p, studenik c r, et al. investigation of the electrophysiological properties of enniatins[j]. archives of biochemistry and biophysics, 2004, 429(2): 215-223.
Properties of ENNIATIN FROM MICROBIAL SOURCE
| storage temp. | 2-8°C |
| solubility | Soluble in DMSO |
| form | powder |
| color | white |
Safety information for ENNIATIN FROM MICROBIAL SOURCE
| Signal word | Danger |
| Pictogram(s) |
 Skull and Crossbones Acute Toxicity GHS06 |
| Precautionary Statement Codes |
P260:Do not breathe dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P312:Call a POISON CENTER or doctor/physician if you feel unwell. |
Computed Descriptors for ENNIATIN FROM MICROBIAL SOURCE
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloropyridazine 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 6-Methyl-2-pyridinemethanol 2-Pyridinecarboxaldehdye tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 1-Indanone 2-Chloromethyl-6-methyl-pyridine 4-Morpholinoaniline Abiraterone Ethyl Ether 1-nitro-3,5-dimethyl adamantine (NMEM) Vardenafil Bis-sulphonamide(Dimer) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran

You may like
-
![3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
2304754-51-4 -
 1126-74-5 99%+View Details
1126-74-5 99%+View Details
1126-74-5 -
 2-Fluoro-6-iodobenzoic acid 99%+View Details
2-Fluoro-6-iodobenzoic acid 99%+View Details
111771-08-5 -
 120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 -
 2-pyridineacetonitrile 2739-97-1 99%+View Details
2-pyridineacetonitrile 2739-97-1 99%+View Details
2739-97-1 -
 4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
138163-08-3 -
![ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
313544-31-9 -
 1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
91419-52-2
