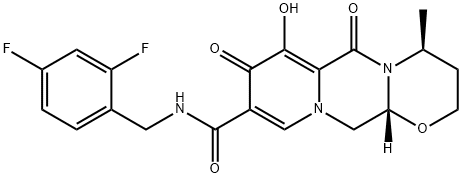
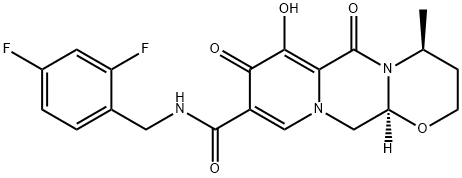
Dolutegravir Impurity 7
- CAS NO.:2244161-71-3
- Empirical Formula: C20H21N3O5
- Molecular Weight: 383.4
- Update Date: 2024-04-22 15:07:34

What is Dolutegravir Impurity 7?
The Uses of Dolutegravir Impurity 7
Desdifluoro Dolutegravir is impurity B of Dolutegravir(D528800), which is a second generation HIV-1 integrase strand transfer inhibitor. Dolutegravir is currently in Phase III clinical trials for the treatment of HIV infection. Dolutegravir has been shown to potently inhibit HIV replication in cells such as peripheral blood mononuclear cells (PBMCs), MT-4 cells and CIP4 cells infected with a self-inactivating PHIV lentiviral vector.
Properties of Dolutegravir Impurity 7
| Boiling point: | 672.0±55.0 °C(Predicted) |
| Density | 1.44±0.1 g/cm3(Predicted) |
| pka | 4.50±1.00(Predicted) |
Safety information for Dolutegravir Impurity 7
Computed Descriptors for Dolutegravir Impurity 7
Dolutegravir Impurity 7 manufacturer
GLP Pharma Standards
3Y
Phone:+91-7893074638
Whatsapp: +91-7893074638
product: Desfluoro Dolutegravir/Dolutegravir Impurity C 2244161-71-3 NLT 95%
New Products
Methyl N-((2'-cyano-[1,1'-biphenyl]-4-yl)methyl)-N-nitroso-L-valinate Levofloxacin - Impurity B/ Levofloxacin Related Compound A Ivabradine R isomer Escitalopram - Impurity K Escitalopram Cyno dilol Impurity Paroxetine EP impurity I/Paroxetine Anhydrous EP Impurity I 2-Amino-5-Bromo-6-Chloropyridine 3-Amino-5-bromopicolinic acid 1-(7,8-Dinitro-4,5-dihydro-1H-1,5-methanobenzo[d]azepin-3(2H)-yl)-2,2,2-trifluoroethanone 2-Amino-3-Bromo-5-Cyanopyridine 2-Amino-3-Chloropyridine 2,5-Dibromo-3-MethylpyridineRelated products of tetrahydrofuran


You may like
-
 Desfluoro Dolutegravir/Dolutegravir Impurity C 2244161-71-3 NLT 95%View Details
Desfluoro Dolutegravir/Dolutegravir Impurity C 2244161-71-3 NLT 95%View Details
2244161-71-3 -
 17471-10-2 Diphenhydramine EP impurity-A /Diphenhydramine Related Compound A NLT 95%View Details
17471-10-2 Diphenhydramine EP impurity-A /Diphenhydramine Related Compound A NLT 95%View Details
17471-10-2 -
 Dorzolamide Related Compound B/Dorzolamide EP Impurity B NLT 95%View Details
Dorzolamide Related Compound B/Dorzolamide EP Impurity B NLT 95%View Details
120279-90-5 -
 76824-16-3 Famotidine - Impurity D NLT 95%View Details
76824-16-3 Famotidine - Impurity D NLT 95%View Details
76824-16-3 -
 118-23-0 NLT 95%View Details
118-23-0 NLT 95%View Details
118-23-0 -
 Miconazole EP Impurity F/ Miconazole USP Related Compound F 47447-55-2 NLT 95%View Details
Miconazole EP Impurity F/ Miconazole USP Related Compound F 47447-55-2 NLT 95%View Details
47447-55-2 -
 2407630-84-4 NLT 95%View Details
2407630-84-4 NLT 95%View Details
2407630-84-4 -
 573704-41-3 NLT 95%View Details
573704-41-3 NLT 95%View Details
573704-41-3
Statement: All products displayed on this website are only used for non medical purposes such as industrial applications or scientific research, and cannot be used for clinical diagnosis or treatment of humans or animals. They are not medicinal or edible.
