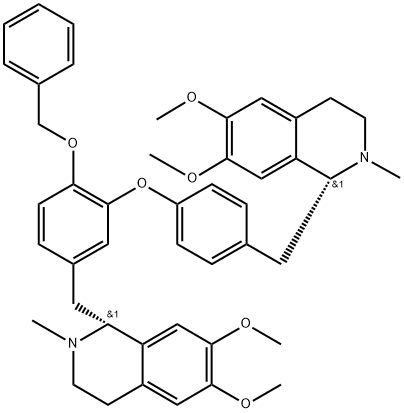
daurisoline
Synonym(s):(-)-Daurisoline;(1R)-1,2,3,4-Tetrahydro-1-[[4-hydroxy-3-[4-[[(1R)-1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-isoquinolinyl]methyl]phenoxy]phenyl]methyl]-6-methoxy-2-methyl-7-isoquinolinol;(R,R)-Daurisoline;O7-Demethyldauricine
- CAS NO.:70553-76-3
- Empirical Formula: C37H42N2O6
- Molecular Weight: 610.74
- MDL number: MFCD00221740
- EINECS: 683-239-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-21 11:20:27
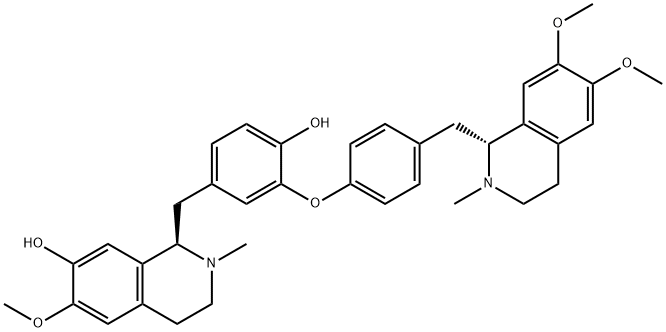
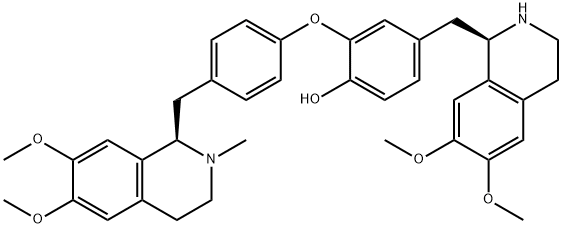
What is daurisoline?
The Uses of daurisoline
Daurisoline is a bisbenzylisoquinoline alkaloid isolated from Menispermum dauricum and displays antiarrythmic effects.
Definition
ChEBI: Daurisoline is a member of isoquinolines.
Properties of daurisoline
| Boiling point: | 724.5±60.0 °C(Predicted) |
| Density | 1.218±0.06 g/cm3(Predicted) |
| storage temp. | -20°C |
| solubility | DMSO: soluble15mg/mL, clear |
| pka | 9.31±0.45(Predicted) |
| form | powder |
| color | white to beige |
Safety information for daurisoline
Computed Descriptors for daurisoline
| InChIKey | BURJAQFYNVMZDV-FIRIVFDPSA-N |
| SMILES | [C@@H]1(CC2=CC=C(O)C(OC3=CC=C(C[C@@H]4C5=C(C=C(OC)C(OC)=C5)CCN4C)C=C3)=C2)C2=C(C=C(OC)C(O)=C2)CCN1C |
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran


You may like
-
 Daurisoline CAS 70553-76-3View Details
Daurisoline CAS 70553-76-3View Details
70553-76-3 -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
Statement: All products displayed on this website are only used for non medical purposes such as industrial applications or scientific research, and cannot be used for clinical diagnosis or treatment of humans or animals. They are not medicinal or edible.
