CYTOCHALASIN J
- CAS NO.:53760-20-6
- Empirical Formula: C28H37NO4
- Molecular Weight: 451.6
- MDL number: MFCD00077708
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 14:57:00
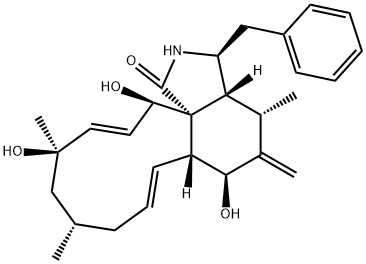
What is CYTOCHALASIN J?
The Uses of CYTOCHALASIN J
Cytochalasin J is a cell-permeable fungal toxin used in actin polymerization studies and cytological research.
What are the applications of Application
Cytochalasin J is a cell-permeable fungal toxin used in actin polymerization studies and cytological research
Biological Activity
cytochalasin j can alter mitotic spindle microtubule organization and kinetochore structure.the cytochalasins are cell-permeable fungal metabolites inhibiting actin polymerization, which can alter diverse processes including cell movement, growth, phagocytosis, degranulation, and secretion.
in vitro
cytochalasin j was identified as a deacetylated analog of cytochalasin h that weakly inhibited actin assembly but significantly altered mitotic spindle microtubule organization and kinetochore structure [1]. in a previous study, cytochalasin j treatment of prometaphase cells reduced the number of kmts and the size and organization of the kinetochore lamina. moreover, in approximately 30% of cells treated with cytochalasin j, the failure of a small number of chromosomes to attach to spindle fibers could be documented. these chromosomes showed a significant change in the organization of the kinetochore laminae. light microscopic analyses of cells treated with cytochalasin j revealed loss of chromosome congression, with chromsomes usually located at the periphery of the spindle. cells treated with 10 microg/ml cytochalasin j for 10 min and released into tissue culture medium showed a resumption of chromosome motion within a few minutes, both during congression and anaphase [2].
Storage
+4°C
References
[1] walling, e. a.,krafft, g.a. and ware, b.r. actin assembly activity of cytochalasins and cytochalasin analogs assayed using fluorescence photobleaching recovery. archives of biochemistry and biophysics 264(1), 321-332 (1988).
[2] wrench, g. a. and snyder, j.a. cytochalasin j treatment significantly alters mitotic spindle microtubule organization and kinetochore structure in ptk1 cells. cell motility and the cytoskeleton 36(2), 112-124 (1997).
Properties of CYTOCHALASIN J
| Melting point: | 138°C (dec.) |
| Boiling point: | 679.5±55.0 °C(Predicted) |
| Density | 1.20±0.1 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | Soluble in ethanol;Soluble in methanol;Soluble in DMSO;Soluble in dimethyl formamide |
| form | white lyophilisate |
| pka | 13.03±0.70(Predicted) |
| color | White |
| Stability: | Light Sensitive |
Safety information for CYTOCHALASIN J
Computed Descriptors for CYTOCHALASIN J
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloropyridazine 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 6-Methyl-2-pyridinemethanol 2-Pyridinecarboxaldehdye tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 1-Indanone 2-Chloromethyl-6-methyl-pyridine 4-Morpholinoaniline Abiraterone Ethyl Ether 1-nitro-3,5-dimethyl adamantine (NMEM) Vardenafil Bis-sulphonamide(Dimer) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran





![[4(N)-3H]CYTOCHALASIN B,CYTOCHALASIN B, [4-3H(N)]-](https://img.chemicalbook.in/StructureFile/ChemBookStructure2/GIF/CB6368649.gif)
You may like
-
![3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
2304754-51-4 -
 1126-74-5 99%+View Details
1126-74-5 99%+View Details
1126-74-5 -
 2-Fluoro-6-iodobenzoic acid 99%+View Details
2-Fluoro-6-iodobenzoic acid 99%+View Details
111771-08-5 -
 120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 -
 2-pyridineacetonitrile 2739-97-1 99%+View Details
2-pyridineacetonitrile 2739-97-1 99%+View Details
2739-97-1 -
 4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
138163-08-3 -
![ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
313544-31-9 -
 1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
91419-52-2
