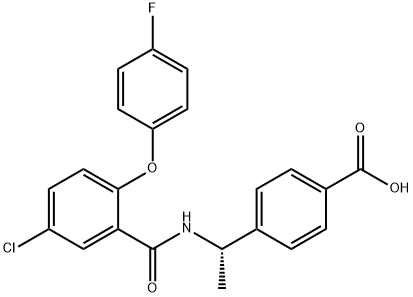
CJ-42794
- CAS NO.:847728-01-2
- Empirical Formula: C22H17ClFNO4
- Molecular Weight: 413.83
- MDL number: MFCD13184815
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-22 18:57:36
What is CJ-42794?
Definition
ChEBI: Benzoic acid, 4-[(1s)-1-[[5-chloro-2-(4-fluorophenoxy)benzoyl]amino]ethyl]- is an aromatic ether.
Biological Activity
ki: 3.16 nm: antagonizes e prostanoid receptor 4 (ep4).ki: 631 nm: antagonizes ep2.cj-42794, as a selective antagonist of ep4, less binds to ep2 and does not have binding affinity for ep1 or ep3. it displays minimal effect on numerous other receptors, channels, or enzymes. cj-42794 delays the healing of gastric ulcers, inhibiting the upregulation of vegf expression and angiogenesis. ep4, activated by prostaglandin e2 (pge2), is a g-protein-coupled receptor, which plays vital roles in bone formation and resorption, cancer, and atherosclerosis via elevating the second messenger cyclic amp (camp).
in vitro
cj-42794 exhibited remarkable binding activity to ep4 and suppressed pge2-triggered elevations of intracellular camp levels in a concentration-dependent fashion in hek293 cells overexpressing human ep4. moreover, cj-42794, concentration-dependently, reversed the inhibitory effects of pge2 on lipopolysaccharide-evoked tumor necrosis factor α production, which suggested that cj-42794 had excellent pharmacological properties used for exploring the physiological role of ep4 [1].
in vivo
male sprague-dawley rats and c57bl/6 mice were given subcutaneously 3 and 10 mg/kg for rats, 10 mg/kg for mice once daily for 7 or 14 days. compared to the controls, cj-42794, in a dose-dependent manner, impaired and delayed the gastric ulcer healing in rats and mice. cj-42794 markedly dampened the increase of vegf expression in ulcerated mucosa of the mouse stomach and the primary gastric fibroblasts of rat [2].
References
[1]. murase, a., taniguchi, y., tonai-kachi, h., nakao, k., & takada, j. in vitro pharmacological characterization of cj-042794, a novel, potent, and selective prostaglandin ep4 receptor antagonist. life sciences. 2008; 82(3-4): 226-232.
[2]. hatazawa, r., tanaka, a., tanigami, m., amagase, k., kato, s., ashida, y., & takeuchi, k. cyclooxygenase-2/prostaglandin e2 accelerates the healing of gastric ulcers via ep4 receptors. ajp: gastrointestinal and liver physiology. 2007; 293(4): g788-g797.
Properties of CJ-42794
| Boiling point: | 572.7±50.0 °C(Predicted) |
| Density | 1.347±0.06 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | insoluble in H2O; ≥11.7 mg/mL in DMSO; ≥54.6 mg/mL in EtOH |
| pka | 4.21±0.10(Predicted) |
| form | A crystalline solid |
| color | White to off-white |
Safety information for CJ-42794
Computed Descriptors for CJ-42794
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran
You may like
-
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 5-Bromo-2-Hydroxy-3-Nitro Pyridine 98%View Details
15862-34-7 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1