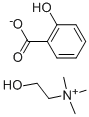
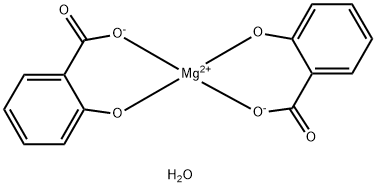
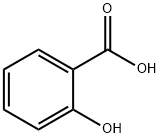
CHOLINE MAGNESIUM TRISALICYLATE
- CAS NO.:64425-90-7
- Empirical Formula: C26H29MgNO10
- Molecular Weight: 539.81
- MDL number: MFCD01746271
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-07-04 15:31:40
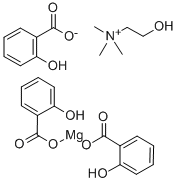
What is CHOLINE MAGNESIUM TRISALICYLATE?
Toxicity
Salicylate intoxication, known as salicylism, may occur with large doses or extended therapy. Common symptoms of salicylism include headache, dizziness, tinnitus, hearing impairment, confusion, drowsiness, sweating, vomiting, diarrhea, and hyperventilation. A more severe degree of salicylate intoxication can lead to CNS disturbances, alteration in electrolyte balance, respiratory and metabolic acidosis, hyperthermia, and dehydration.
The Uses of CHOLINE MAGNESIUM TRISALICYLATE
The antipyretic and analgesic effect of Choline magnesium trisalicylate is similar to that of aspirin, but the adverse reaction is less than that of aspirin, and the curative effect is better than that of aspirin and ibuprofen. It is indicated for the relief of symptoms of rheumatoid arthritis, osteoarthritis, and other arthritic conditions, both in the acute onset and in the long-term treatment of these diseases. The gastrointestinal reaction is small, so it is especially suitable for patients who cannot tolerate aspirin.
Background
Choline magnesium trisalicylate is a non-acetylated salicylate used widely as a nonsteroidal anti-inflammatory drug. Trisalicylate significantly reduces methotrexate renal clearance, displacing methotrexate from protein, increasing the fraction unbound by 28% .
Indications
Choline magnesium trisalicylate is used to reduce pain and inflammation caused by conditions such as arthritis. This medication is also used to treat fever in adults.
Pharmacokinetics
Trisalicylate-choline is a non-steroidal anti-inflammatory drug (NSAID) that contains a combination of choline salicylate and magnesium salicylate. Does not affect platelet aggregation.
Side Effects
①Active peptic ulcer, hemophilia and allergic to salicylates should not be used. Pregnant women and children under 12 years old are prohibited. ②Use with caution in patients with chronic renal insufficiency and acute erosive gastritis. ③ This product should not be used at the same time as coumarin anticoagulant or heparin. Concomitant use with steroids or alcohol may increase ulcer bleeding. ④ Occasionally rash, fever and stomach discomfort.
Metabolism
Not Available
Safety information for CHOLINE MAGNESIUM TRISALICYLATE
New Products
2-(2-aminoethyl) benzenesulfonamide Gemcitabine 3-benzoate impurity Dihydroxyphenylacetone Ornidazole EP impurity D Acetyl amino diol impurity Valsartan Benzyl Ester Impurity B N,N'-Dicyclohexylcarbodiimide 3-Bromo-2-Methyl-5-Nitropyridine 5-Bromo-3-Methyl-2-Pyridinecarboxylic acid 2-Amino-4-Methylpyridine 4-Aminopyridine 4-dimethylaminopyridineRelated products of tetrahydrofuran

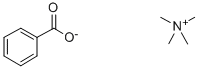
You may like
-
 4-(Methylnitrosoamino)-butanal NLT 95%View Details
4-(Methylnitrosoamino)-butanal NLT 95%View Details
339362-86-6 -
 848696-99-1 NLT 95%View Details
848696-99-1 NLT 95%View Details
848696-99-1 -
 Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
1384752-15-1 -
 615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 -
 175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 -
 S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
1346605-15-9 -
 Valsartan Related Compound A NLT 95%View Details
Valsartan Related Compound A NLT 95%View Details
137862-87-4 -
 Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
124750-49-8
