BENZILIC ACID 1-ETHYLPIPERIDIN-3-YL ESTER METHOBROMIDE
- CAS NO.:125-51-9
- Empirical Formula: C22H28BrNO3
- Molecular Weight: 434.37
- MDL number: MFCD00079275
- EINECS: 204-741-2
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-04 15:05:47
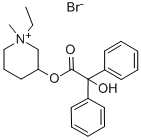
What is BENZILIC ACID 1-ETHYLPIPERIDIN-3-YL ESTER METHOBROMIDE?
Originator
Piptal ,Merrell National ,US ,1955
The Uses of BENZILIC ACID 1-ETHYLPIPERIDIN-3-YL ESTER METHOBROMIDE
antispasmodic
The Uses of BENZILIC ACID 1-ETHYLPIPERIDIN-3-YL ESTER METHOBROMIDE
Piptalake is used in the treatment with copper-tolylterpyridine and platinum-tolylterpyridine that targets telomeres and telomerase for regulation of chromosome mis-segregation rates for cancer therapy.
Manufacturing Process
N-ethyl-3-chloropiperidine was prepared according to the method of Fuson and Zirkle described in Volume 70, J. Am. Chem. Soc., p 2760. 12.0 g (0.081 mol) of N-ethyl-3-chloropiperidine was mixed with 18.6 g (0.081 mol) of benzilic acid and 80 cc of anhydrous isopropyl alcohol as a solvent. The mixture was refluxed for 72 hours. The solution was then filtered and concentrated at 30 mm of mercury. The concentrate was dissolved in water, acidified with hydrochloric acid and extracted with ether to remove theThe aqueous layer was neutralized with sodium bicarbonate and the product was extracted with ether. The ethereal solution of the product was dried with potassium carbonate, the ether was removed by distillation and the residue was distilled at 0.12 to 0.18 mm of mercury, the BP being 194° to 198°C. A yield of 16.5 g (60% of theoretical) of N-ethyl-3-piperidyl-benzilate was obtained.
34 g (0.1 mol) of the basic ester is dissolved in 75 cc of isopropyl alcohol and treated with 9.5 g (0.1 mol) of methyl bromide. The mixture is allowed to stand at room temperature until precipitation is complete. The product is removed by filtration and washed with isopropyl alcohol, yield 33 g, MP 175° to 177°C. On recrystallization from isopropyl alcohol, the MP was raised to 179° to 180°C dec.
brand name
Piptal (Marion Merrell Dow).
Therapeutic Function
Spasmolytic
Properties of BENZILIC ACID 1-ETHYLPIPERIDIN-3-YL ESTER METHOBROMIDE
| Melting point: | 179-180° |
| form | powder |
| color | white |
Safety information for BENZILIC ACID 1-ETHYLPIPERIDIN-3-YL ESTER METHOBROMIDE
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral |
Computed Descriptors for BENZILIC ACID 1-ETHYLPIPERIDIN-3-YL ESTER METHOBROMIDE
New Products
Levothyroxine Impurity-F Montelukast EP Impurity-D/Montelukast USP Related Compound C Atorvastatin FXA Impurity/Atorvastatin Cyclic 6-Hydroxy Impurity Sodium Salt Isosulfan blue Keto N-Oxide Impurity Ivermectin Impurity F N-Nitroso des Methyl Tramadol/N-Nitroso-N-Desmethyl-Tramadol DL-beta-(3-Bromophenyl)alanine Tetrabutylammonium perchlorate N,O-Dimethylhydroxylamine hydrochloride (RS)-beta-Amino-beta-(4-bromophenyl)propionic acid 2-Amino-5-bromo-4-(trifluoromethyl)pyridine(RM for Indian lab) N,N CARBONYL DIIMIDAZOLE (R)-BoroLeu-(+)-Pinanediol-CF3COOH 1,4-bis(methylsulfonyl)butane 4-(5-amino-1-methyl-1h-benzoimidazol-2-yl)-butyric acid isopropyl ester. 5-Methyl-1,3-benzenediacetonitrile 4-Fluorothiophenol 1-methyl amino-2,4-dinitro benzene CSA (DL-10-Camphorsulfonic Acid) 2-Hydroxy-4-methylnicotinic acid 3-iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine (2S)-1-((2S,3S)-3-(2-methylbutyl)-4-oxooxetan-2-yl)pentadecan-2-yl formylleucinate 1-(3,5-dichlorophenyl)-2,2,2-trifluoroethane-1-sulfonyl chloride S-(1-(3,5-dichlorophenyl)-2,2,2-trifluoroethyl) ethanethioateRelated products of tetrahydrofuran
You may like
-
 674783-97-2 98+View Details
674783-97-2 98+View Details
674783-97-2 -
 H-D-TRP(FOR)-OH HCL 98+View Details
H-D-TRP(FOR)-OH HCL 98+View Details
367453-01-8 -
 DL-beta-(3-Bromophenyl)alanine 117391-50-1 98+View Details
DL-beta-(3-Bromophenyl)alanine 117391-50-1 98+View Details
117391-50-1 -
 1923-70-2 Tetrabutylammonium perchlorate 98+View Details
1923-70-2 Tetrabutylammonium perchlorate 98+View Details
1923-70-2 -
 49830-37-7 98+View Details
49830-37-7 98+View Details
49830-37-7 -
 3-Amino-3-(4-fluorophenyl)propanoic acid 98+View Details
3-Amino-3-(4-fluorophenyl)propanoic acid 98+View Details
325-89-3 -
 1428243-26-8 98+View Details
1428243-26-8 98+View Details
1428243-26-8 -
 1-aminocyclopentane carbonitrile, HCl 98+View Details
1-aminocyclopentane carbonitrile, HCl 98+View Details
16195-83-8
